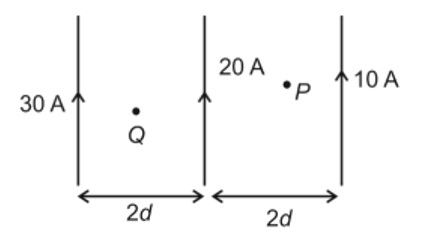
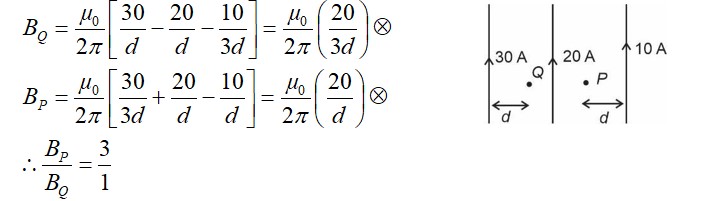Number of paramagnetic oxides among the following given oxides is_________.
Li2O, CaO, Na2O2, KO2, MgO and K2O
Number of paramagnetic oxides among the following given oxides is_________.
Li2O, CaO, Na2O2, KO2, MgO and K2O
Given are the oxide of alkali and alkaline earth metals which are ionic in nature.
Simple oxide are Li2O, CaO, MgO and K2O.
Peroxide is Na2O2 and superoxide is KO2.
All simple oxides are diamagnetic as it has no unpaired electron.
Similar Questions for you
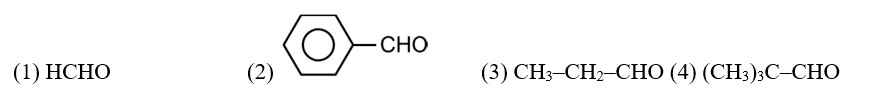
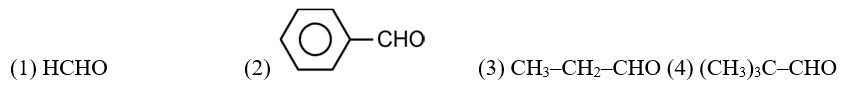
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Acetaldehyde (CH3CHO) gives positive lodoform test and positive Fehling's solution test
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering