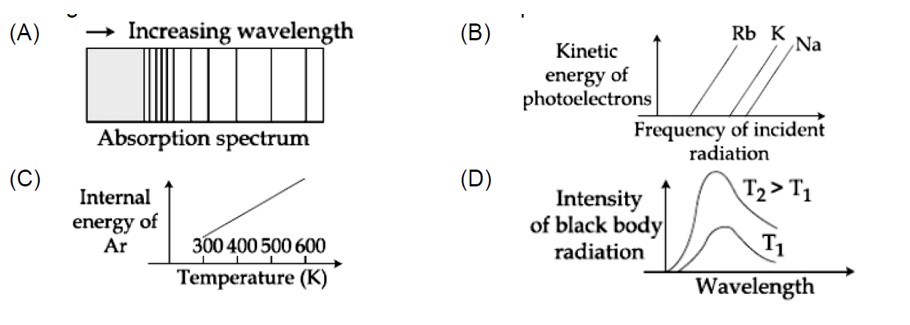
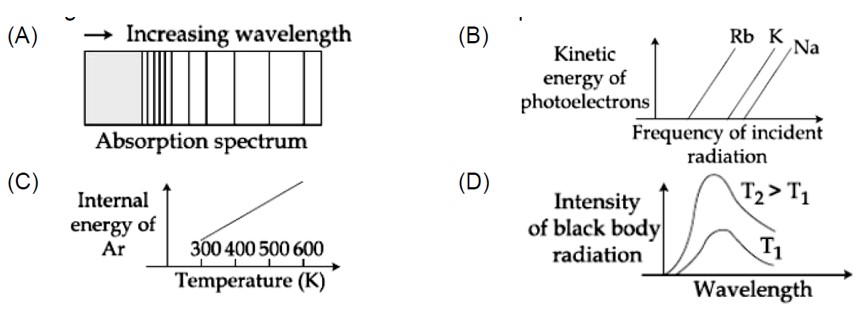
The figure that is not a direct manifestation of the quantum nature of atom is:
The figure that is not a direct manifestation of the quantum nature of atom is:
Similar Questions for you
1, 2 and 3 are according to quantum theory but (4) is statement of kinetic theory of gases
Through Heisenberg's Uncertainty Principle, it's proven that you can't pin down where an electron is and how fast it's moving at the same time. The Bohr model did not look into this. The main reason for that thought was that it pictured electrons like little planets that would move in a loop around
De Broglie had the idea that everything that moves has a bit of wave behaviour. Technically, that includes any person, a football, and even a bus. The catch is that for big things, the mass is so large that their wavelength is insanely tiny. It's so tiny it's impossible to notice. That's
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering