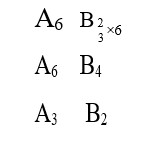The gas phase reaction 2A(g) ? A?(g) At 400 K has ΔG° = +25.2 kJ mol?¹.
The equilibrium constant K? for this reaction is ________ *10?². (Round off to the nearest integer).
[ Use: R = 8.3 J mol?¹ K?¹, In10 = 2.3, log?? 2 = 0.30, 1atm = 1 bar]
[antilog (-0.3) = 0.501]
The gas phase reaction 2A(g) ? A?(g) At 400 K has ΔG° = +25.2 kJ mol?¹.
The equilibrium constant K? for this reaction is ________ *10?². (Round off to the nearest integer).
[ Use: R = 8.3 J mol?¹ K?¹, In10 = 2.3, log?? 2 = 0.30, 1atm = 1 bar]
[antilog (-0.3) = 0.501]
ΔG° = + 25.2 kJ / mol
Using ΔG° = -2.3 RT log Kp
25.2 * 10³ = - 2.3 * 8.3 * 400 log Kp
o 3.3 = log Kp
log (1 / 2*10³) = log Kp
Kp = 1 / (2*10³)
Using; Kp = Kc (RT)
1 / (2*10³) = Kc (0.083 * 400)? ¹
Similar Questions for you
CH3COOH + NaOH → CH3COONa + H2O
ΔH = –50.6 kJ/mol
NaOH + SA [HCl] → NaCl + H2O
ΔH = –55.9 kJ/mol
the value of ΔH for ionisation of CH3COOH
⇒ ΔH = +55.9 – 50.6
5.3 kJ/mol
Kindly consider the solution
Fact.
Kindly go through the solution
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering