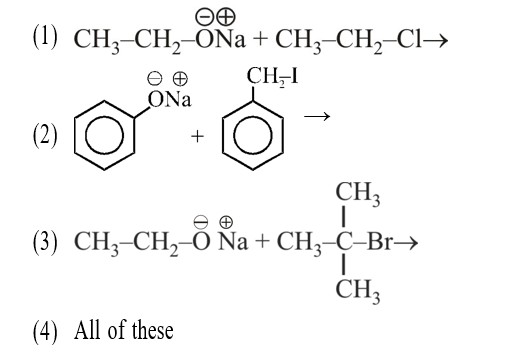
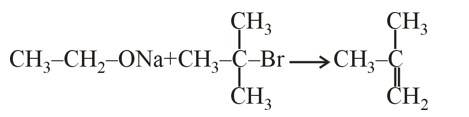
The reaction that occurs in a breath analyser, a device used to determine the alcohol level in a person's blood stream is
If the rate of appearance of Cr2(SO4)3 is 2.67 mol min-1 at a particular time, the rate of disappearance of C2H6O at the same time is________mol min-1. (Nearest integer)
The reaction that occurs in a breath analyser, a device used to determine the alcohol level in a person's blood stream is
If the rate of appearance of Cr2(SO4)3 is 2.67 mol min-1 at a particular time, the rate of disappearance of C2H6O at the same time is________mol min-1. (Nearest integer)
Given that, rate of appearance of Cr2(SO4)3 =
LHS RHS
Rate of disappearance of C2H6O =
Rate of disappearance of C2H6O = 4.005 mol /min
Ans. = 4 (nearest integer)
Similar Questions for you
ΔG° = –RT * 2.303 log K
–nFE° = +RT * 2.303 log K
2 * 96500 * 0.295 = 8.314 * 298 * 2.303 log10 K
10 = log10 K = 1010
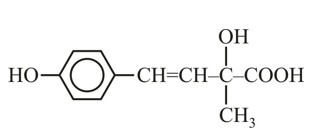
It has chiral centre and differently di substituted double bonded carbon atoms.
Rate of ESR ∝ No. of α – H (Hyperconjugation)
Cr3+ion is a most stable in aqueous solution due to. t2g half filled configuration
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering