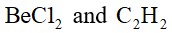1 Answer
E
Answered by
9 months ago
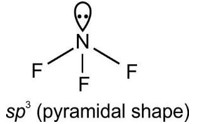
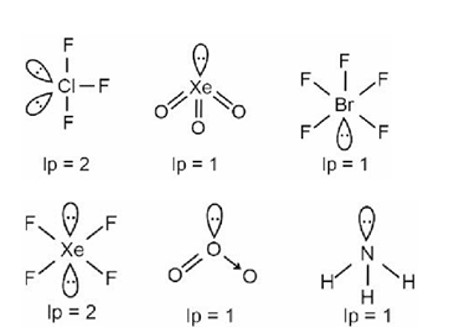
The Valence Shell Electron Pair Repulsion (VSEPR) Theory is an algorithm developed to predict the molecular geometry of the compounds. The VSEPR theory predict the molecular shape based on the repulsion between electron pairs (bonding and lone) around the central atom. As per the NCERT Textbooks:
“Ac
Similar Questions for you
He2 has zero bond order hence it does not exist.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Learn more about...

Chemistry Chemical Bonding and Molecular Structure 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering