Which acid of each pair shown here would you expect to be stronger?
(i) CH3CO2H or CH2FCO2H
(ii) CH2FCO2H or CH2ClCO2H
(iii) CH2 FCH2 CH2 CO2H or CH3 CHFCH2 CO2H
Which acid of each pair shown here would you expect to be stronger?
(i) CH3CO2H or CH2FCO2H
(ii) CH2FCO2H or CH2ClCO2H
(iii) CH2 FCH2 CH2 CO2H or CH3 CHFCH2 CO2H
(i) The +I effect of –CH3 group increases the electron density on the O-H bond. Therefore, the release of proton becomes difficult. On the other hand, the -I effect of F decreases the electron density on the O-H bond. Therefore, the proton can be released easily. Hence, CH2FCO2H is a stronger acid
Similar Questions for you
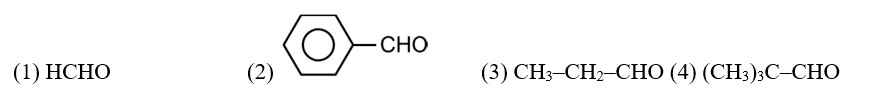
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Acetaldehyde (CH3CHO) gives positive lodoform test and positive Fehling's solution test
CH3—CH2—CHO does not undergo Cannizzaro reaction because it has α-hydrogen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Aldehydes, Ketones and Carboxylic Acids 2021
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering