11.22 Answer the following questions based on the P – T phase diagram of CO2:
(a) CO2 at 1 atm pressure and temperature – 60 °C is compressed isothermally. Does it go through a liquid phase ?
(b) What happens when CO2 at 4 atm pressure is cooled from room temperature at constant pressure ?
(c) Describe qualitatively the changes in a given mass of solid CO2 at 10 atm pressure and temperature –65 °C as it is heated up to room temperature at constant pressure.
(d) CO2 is heated to a temperature 70 °C and compressed isothermally. What changes in its properties do you expect to observe?
11.22 Answer the following questions based on the P – T phase diagram of CO2:
(a) CO2 at 1 atm pressure and temperature – 60 °C is compressed isothermally. Does it go through a liquid phase ?
(b) What happens when CO2 at 4 atm pressure is cooled from room temperature at constant pressure ?
(c) Describe qualitatively the changes in a given mass of solid CO2 at 10 atm pressure and temperature –65 °C as it is heated up to room temperature at constant pressure.
(d) CO2 is heated to a temperature 70 °C and compressed isothermally. What changes in its properties do you expect to observe?
11.22
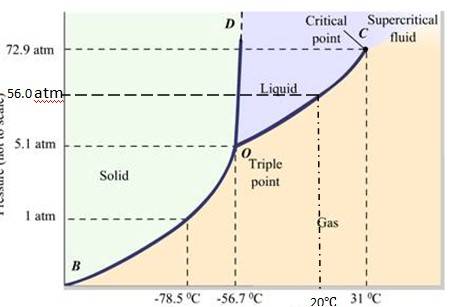
The fusion and boiling points are given by the intersection point where this parallel line cuts the fusion and vaporization curves. It departs from ideal gas behavior as pressure increases.
(a) At 1 atm pressure and at – 60 °C, it lies left of -56.6 (triple point O). Hence, it lies in the regi
Similar Questions for you
According to question, we can write
Heat Released by block = Heat gain by large Ice block
5 × 0.39 × 500 = mice × 335
= 2.91 kg
Given
6a2 = 24
a2 = 4
a = 2m
This is a multiple choice answer as classified in NCERT Exemplar
(b), (c), (d) When the hot milk in the table is transferred to the surroundings by conduction, convection and radiation.
According to newton's law of cooling temperature of the milk falls of exponentially. Heat also will be transferred
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

physics ncert solutions class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering