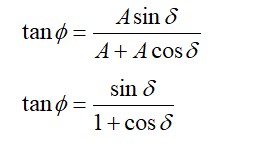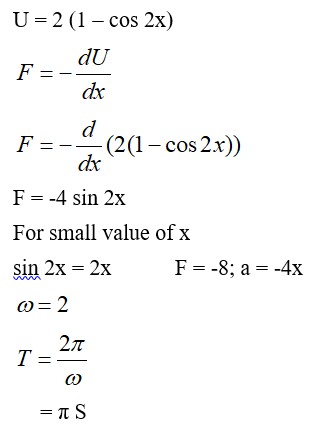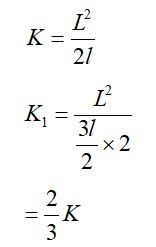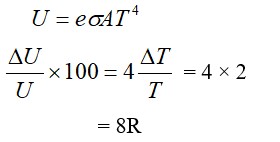An ideal gas in a closed container is slowly heated. As its temperature increases, which of the following statements are true?
(a) the mean free path of the molecules decreases
(b) the mean collision time between the molecules decreases.
(c) the mean free path remains unchanged.
(d) the mean collision time remains unchanged.
An ideal gas in a closed container is slowly heated. As its temperature increases, which of the following statements are true?
(a) the mean free path of the molecules decreases
(b) the mean collision time between the molecules decreases.
(c) the mean free path remains unchanged.
(d) the mean collision time remains unchanged.
On increasing the temperature, random velocity of molecules increases, therefore mean collision time between the molecules decreases. But the mean free path remains constant as it is product of velocity and time.? (b) and (c) are correct.
Similar Questions for you
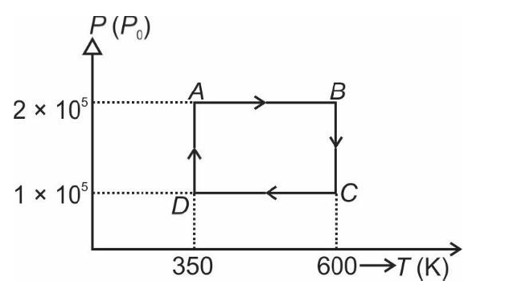
From A to B the process is isobaric
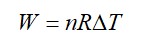
= W = 2 × R (600 - 350)
= 500 R
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Physics Units and Measurement 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering