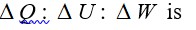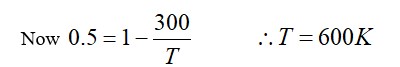Consider that an ideal gas (n moles) is expanding in a process given by P = f ( V ), which passes through a point (Po, V0 ). Show that the gas is absorbing heat at (P0, Vo ) if the slope of the curve P = f (V ) is larger than the slope of the adiabat passing through (Po, V0 ).
Consider that an ideal gas (n moles) is expanding in a process given by P = f ( V ), which passes through a point (Po, V0 ). Show that the gas is absorbing heat at (P0, Vo ) if the slope of the curve P = f (V ) is larger than the slope of the adiabat passing through (Po, V0 ).
This is a long answer type question as classified in NCERT Exemplar
Slope of the curve = f(V) , where V is the volume
Slope of P = f(V) curve at ((Po, V0 )= f(Vo)
Slope of adiabatic at (Po, V0 )= k(-Y)Vo-1-Y =-YPo/Vo
Now heat absorbed in the process P= f(V)
dQ=dU+dW= nCvdT+pdV
pV=nRT
T= pV/nR
T=
nCv
A
Similar Questions for you
From A to B the process is isobaric
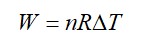
= W = 2 × R (600 - 350)
= 500 R
Heat is path dependent so path function but internal energy does not depend on path chosen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

physics ncert solutions class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering