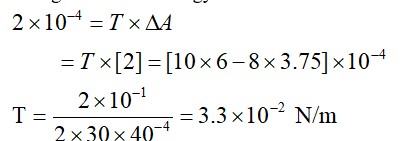How did Rutherford know that an atom was mostly empty space?
How did Rutherford know that an atom was mostly empty space?
In the gold foil scattering experiment by Rutherford and his team, almost all alpha particles passed straight through the foil. There were minor deflections mostly, and only a small amount of them showed deflections at really large angles. Only a few of them rebounded. This was the main observation
Similar Questions for you
In Rutherford's model, electrostatic force provides the centripetal force:
∈
Kinetic Energy (K):
Potential Energy (U):
Total Energy (E):
Note that, E is the Epsilon symbol.
Based on what classical electromagnetic theory says, we know that an accelerated charged particle must radiate energy or electromagnetic waves. That remains continuous.
By looking at the Rutherford’s atomic model we can assume that electrons would revolve around the nucleus, which in this logic
Kindly go through the solution
Change in surface energy = work done
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Physics Atoms 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering