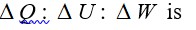One mole of an ideal gas is taken through an adiabatic process where the temperature rises from 27°C to 37°C. If the ideal gas is composed of polyatomic molecule that has 4 vibrational modes, which of the following is true? [R = 8.314 J mol⁻¹K⁻¹]
One mole of an ideal gas is taken through an adiabatic process where the temperature rises from 27°C to 37°C. If the ideal gas is composed of polyatomic molecule that has 4 vibrational modes, which of the following is true? [R = 8.314 J mol⁻¹K⁻¹]
Degrees of freedom (f) = 3 translational + 3 rotational + (2 * 4) vibrational = 14
γ = 1 + 2/f = 1 + 2/14 = 8/7
W = nR? T/ (γ-1) = (1 * 8.3 * (310-300)/ (8/7 - 1) = (83)/ (1/7) = 581 J
As W is positive, work is done by the gas. The solution says W<0, work done on the gas. This implies? T is negative. The question states temperature rises, so work is done on the gas. W = -582J.
Similar Questions for you
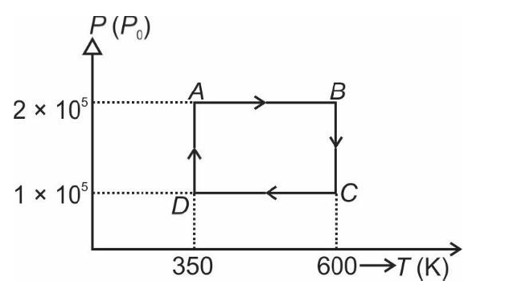
From A to B the process is isobaric
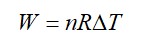
= W = 2 × R (600 - 350)
= 500 R
Heat is path dependent so path function but internal energy does not depend on path chosen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering