Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 9
DES Pune University offers BTech and MBA programmes to students as its top courses. The college offers these programmes to students at UG and PG levels. DES offers these programmes for a duration of 4 and 2 years, respectively with a comprehensive curriculum. Further, DES PU courses are offered to students in full-time mode and are taught by highly educated faculty members.
New Question
10 months agoContributor-Level 10
There are more than 30 BCA colleges in Thrissur. Students can pursue a BCA course at 15 private and 8 public-private colleges. They can take admission to these colleges based on their score in Class 12.
New Question
10 months agoContributor-Level 10
The S.P. Group of Colleges provide good placements to its graduating MBA students. The students are given excellent placement support every year placing them in top companies. Check out the table below to know the key highlights of the S.P. Group of Colleges placement between 2021- 2023:
Particulars | Placement Statistics (2021) | Placement Statistics (2022) | Placement Statistics (2023) |
|---|---|---|---|
the highest Package | INR 3 LPA | INR 3.25 LPA | INR 4.2 LPA |
Average Package | INR 2.4 LPA | INR 2.4 LPA | INR 3.2 LPA |
Students Placed | 155 | 327 | 451 |
Total Recruiters | 8 | 10 | 14 |
Top Sector | PSU's | Pharmacy | Agriculture |
Placement Rate | 60% | 65% | 71% |
Note- the placement report for 2024 is yet to be released.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
It's worth noting that when the temperature drops, the values of Henry's law constant (KH) rise. Because of this, the solubility of oxygen in water increases with decreasing temperature at a given pressure. As a result, the presence of more oxygen at lower temperatures makes aquatic organisms feel more at ease in cold water than in warm water.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (ii) i.e., It has low concentration of oxidizing agents is the answer since the common components of photochemical smog are ozone, nitric oxide, acrolein, formaldehyde and peroxyacetyl nitrate (PAN). Photochemical smog causes serious health problems and both ozone and PAN act as powerful eye irritants. Photochemical smog contains high concentrations of oxidants.
New Question
10 months agoContributor-Level 10
We have, f(x) =
{
At
So we have three disjoint internal i.e.,
When,
So, f(x) is increasing.
When
So, f(x) is decreasing.
When
f(x) =
So, f(x) is increasing.
f(x) is increasing for x(∞,1) and [1, ∞] and decreasing for x[1, 1].
New Question
10 months agoContributor-Level 10
Yes, admissions are open at BNM Institute of Technology. Follow the BNM Institute of Technology application process mentioned below:
Step 1: Visit the official website of BNM Institute of Technology.
Step 2: Select the required course.
Step 3: Pay the application fee.
Step 4: Fill out and submit the application form with the required information.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
p =KHx (where p is the partial pressure of the gas in the vapour phase and x is the mole fraction of the gas in solution) is Henry's law expressed mathematically.
As a result of the aforementioned equation, "the lower the solubility of the gas in the liquid, the greater the value of Henry's law constant KH at a given pressure."
New Question
10 months agoContributor-Level 10
The S.P. Group of Colleges are known for its excellent placement record. Check out the tabulated data given below to know more about S.P. Group of Colleges placements in 2021-2023:
Particulars | Placement Statistics (2021) | Placement Statistics (2022) | Placement Statistics (2023) |
|---|---|---|---|
the highest Package | INR 3 LPA | INR 3.25 LPA | INR 4.2 LPA |
Average Package | INR 2.4 LPA | INR 2.4 LPA | INR 3.2 LPA |
Students Placed | 155 | 327 | 451 |
Total Recruiters | 8 | 10 | 14 |
Top Sector | PSU's | Pharmacy | Agriculture |
Placement Rate | 60% | 65% | 71% |
Note- the placement report for 2024 is yet to be released.
New Question
10 months agoContributor-Level 10
We have, f(x)=
So,
Now,
And, as cos x lies in [1, 1].
So, (i) for increasing, f(x) ≥ 0.
cosx ≥ 0.
x lies in Ist and IVth quadrant.
i.e., f(x) is increasing for and
(ii) for decreasing, f(x) ≤ 0.
cosx ≤ 0.
x lies in IInd and IIIrd quadrant.
i.e., f(x) is decreasing for .
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (iii)
i.e., Decrease in the amount of dissolved oxygen in water is the answer since organic waste is oxidized by microorganisms in the presence of dissolved oxygen. Hence, oxygen decreases in water as a result it is harmful for aquatic life.
New Question
10 months agoContributor-Level 10
BNM Institute of Technology is a good college. BNM Institute of Technology is accredited with an A Grade by NAAC. Most of the UG branches are accredited by the NBA. The college has a total of 10 departments and has 12 centres of excellence, which are known to serve as innovation hubs, equipping various students with cutting-edge skills, real-world exposure, and several opportunities for breakthrough developments. The college's reputation is really good.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The number of moles of solute dissolved in one litre of solution is the molarity of a solution, which is defined as "the number of moles of solute dissolved in one litre of solution." Because volume is affected by temperature and changes with it, the molarity will also change as the temperature changes.
Other concentration words, such as mass percentage, ppm, mole fraction, and molality, are based on the mass-to-mass relationship of the solute and solvent in a binary solution. Because mass does not vary as a function of temperature, these concentration terms do not chan
New Question
10 months agoContributor-Level 10
Vishwakarma University course admissions are based on entrance exam score, as per the course requirement. Vishwakarma University course admission is subject to fulfilment of the admission criteria. The university provides a wide range of programmes across multiple specialisations. Admissions to the programmes offered at the university are primarily entrance-based. Students can check course-wise selection criteria below:
Course | Selection Criteria |
|---|---|
BA | Merit-Based |
BE/ BTech | VUNET/ MH – CET / JEE Mains |
BDes | VUNET/UCEED |
MBA | VUNET/ MH-CET |
MSc | Merit-Based |
New Question
10 months agoContributor-Level 10
We have
Slope of the normal at any point is
The equation of the normal at a given point is given by,
Now, the perpendicular distance of the normal from the origin is
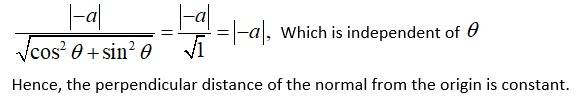
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The solubility rule "like dissolves like" is based on the intermolecular forces of that exist in solution as follows:
If the intermolecular interactions in both components are similar, a substance (solute) dissolves in a solvent (ie. solvent and solute particles or molecules). When polar solutes dissolve in polar solvents and non-polar solutes dissolve in non-polar solvents, this is a regular occurrence.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (i) i.e., Ozone is not responsible for greenhouse effect is the answer since O3 is responsible for the greenhouse effect, its contribution is about 8% to 10%. About 75% of the solar energy reaching the earth is absorbed by the earth's surface, which increases its temperature. The rest of the heat radiates back to the atmosphere while some of the heat is trapped by gases such as carbon dioxide, methane, ozone, chlorofluorocarbon compounds (CFCs) and water vapour in the atmosphere. Thus, they add to the heating of the atmosphere. This is the reason behind glo
New Question
10 months agoContributor-Level 10
No, the MIT School of Vedic Sciences, MIT- ADT University BA fee structure differs for NRI, PIO, OCI, and foreign nationals. These candidates are required to pay 1.5 times the tuition, development, and university fees that Indian students pay. This fee adjustment helps the university accommodate the additional administrative and resource needs associated with international admissions and student services.
New Question
10 months agoContributor-Level 10
Equation of the curve is
Now, the slope of the normal at point is
Equation of the normal at is
New Question
10 months agoContributor-Level 10
Yes, the Shri Vishwakarma Skill University offers BTech programmes in various specialisations. It is a four-year full-time programme, which is divided into eight semesters. Candidates can refer to the following key highlights for more information:
| Particular | Highlights |
|---|---|
| Duration | Four years |
| Semesters | Eight |
| Mode | Full-time |
| Level | UG-level |
| Seat Intake | 180 |
| Selection | Entrance-based |
| Tuition Fee | INR 1.8 Lacs to INR 2.7 lakh |
NOTE: The mentioned fee and seat intake is taken from the official website/ sanctioning body. It is still subject to change and, hence, is indicative.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
