Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The vapour pressure of a liquid in comparison to air pressure determines its boiling point. At a constant atmospheric pressure, the lower the vapour pressure, the higher the boiling point of a liquid, and vice versa.
Because NaCl is a nonvolatile solute, it reduces the vapour pressure of water when added to it. The boiling point of water rises as a result. Methyl alcohol, on the other hand, is more volatile than water, therefore adding it to the solution raises the overall vapour pressure, lowering the boiling point of water.
New Question
10 months agoContributor-Level 10
Let ‘b’ and ‘x’ be the fixed base and equal side of isosceales triangle.
Then, cm/s (Ø decreasing).

New Question
10 months agoContributor-Level 10
Admission to the BA (Hons) Psychology and Philosophy & Yoga programmes at MIT School of Vedic Sciences, MIT-ADT University is purely merit-based. Students are shortlisted based on their Class 12 marks, with a minimum eligibility requirement of 50% or equivalent. No entrance exam is conducted for these courses, and selection is made through a merit list followed by document verification and partial fee payment.
New Question
10 months agoContributor-Level 10
Candidates willing to get admission must take part in the BNMIT admission process before the last date of application. Candidates must know their BNMIT eligibility criteria and apply accordingly. Follow the BNM Institute of Technology application process mentioned below:
Step 1: Visit the official website of BNM Institute of Technology.
Step 2: Select the required course.
Step 3: Pay the application fee.
Step 4: Fill out and submit the application form with the required information.
New Question
10 months agoContributor-Level 10
Because both components exist in the distillate and the liquid and vapour compositions are the same, this indicates that the liquids have formed an azeotropic combination that cannot be separated at this stage by fractional distillation.
New Question
10 months agoContributor-Level 10
We have, f(x)
f(x) =
f(x) =
=
= =
At extreme points, f(x) = 0.
At x = e, f"(e) =
x = e is a point of maximum.
New Question
10 months agoContributor-Level 10
There are several factors that Taxila Business School considers while releasing the cutoff for the PGDM course. Some of the factors that the school considers before releasing the cutoff are the number of applicants, scores candidates have achieved in the entrance examination, number of available seats, previous years' cutoff trends, etc.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (i) i.e., rich in dissolved oxygen is the answer since the total amount of oxygen consumed by microorganisms in decomposing organic matter present in a certain volume of water is known as Biochemical Oxygen Demand (BOD) of water. If the BOD value is less than 5 ppm, then the sample of water is considered to be pure and is rich in dissolved oxygen.
New Question
10 months agoContributor-Level 10
BNM Institute of Technology is an autonomous college under the VTU and approved by AICTE (All India Council for Technical Education). B.N.M Educational Institutions consist of BNM Montessori House, BNM Primary School (State Syllabus), BNM High School (State Syllabus), BNM Public School (Central Syllabus), BNM PU College, BNM Degree College, and BNM Institute of Technology. The college provides UG, PG, and PhD programmes to students. These courses are provided across the BTech/ Management, and various other streams. The flagship programmes available at this college are BTech/ B.E. and&
New Question
10 months agoContributor-Level 10
Students who wish to take admission into the Shri Guru Ram Rai University courses must fill out an application form. After filling out the form, they are required to pay an application fee of INR 1,000. This fee is non-refundable under any circumstances.
Note: The above-mentioned fee is as per official sources. However, it is subject to change.
New Question
10 months agoContributor-Level 10
(a) Consider
Now, is approximately equal to and is given by,
Hence, the approximate value of is
=0.677
(b)
(b) Consider
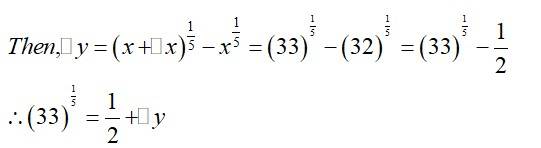
Now, is approximately equal to and is given by,
Hence, the approximate value of
is
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
When dissolved in suitable solvents, certain solutes/compounds either dissociate or associate. For instance, ethanoic acid dimerises in benzene due to hydrogen bonding, but dissociates and produces ions in water.
As a result, the number of chemical species in solution rises or falls in relation to the number of chemical species of solute used to create the solution. Because the magnitude of the colligative property is dependent on the number of solute particles in relation to the total number of particles in solution, it is expected that the molar mass calculated using c
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (i)
i.e., Its main components are produced by the action of sunlight on emissions of automobiles and factories is the answer since classical smog is a mixture of smoke, fog and sulphur dioxide and occurs in cold humid climates. While the gases released by automobiles and factories are not responsible for classical fog.
New Question
10 months agoContributor-Level 10
We have,
At f(x) = 0.
2x – 1 = 0
=
Option (B) is
Hence maximum value of f(x) = at x = 1 and x = 0.
Option (c) is correct.
New Question
10 months agoContributor-Level 10
Vishwakarma University course admission is subject to fulfilment of the admission criteria. The university provides a wide range of programmes across multiple specialisations. Admissions to the programmes offered at the university are primarily entrance-based. Students can check the table below to know some of the detailed course-wise eligibility:
Course | Eligibility |
|---|---|
BA | Class 12 with English as a passing subject or 3 years Diploma or equivalent from a recognised Board. |
BE/ BTech | Class 12 with English, Physics, and Mathematics as compulsory subjects with a minimum 45% aggregate (40% for reserved category) |
BDes | Class 12 with English as a passing subject minimum 45% aggregate (40% for reserved category) or DVoc stream in the same or allied sector |
MBA | Bachelor's with minimum 50% aggregate (45% for reserved category) |
MSc | BE / BTech / BSc in CSE / ECE / EEE / ISE / TE / BCA qualification with 50% (45% for reserved category) marks from any recognised University. Preferred from respective backgrounds. |
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
(i) When egg is placed in a dilute mineral acid solution (preferably dilute HCl solution), the hard external CaCO3 layer of the egg dissolves out /removed without damaging its semipermeable membrane.
(ii) Yes, this egg can be inserted into a bottle with a narrow neck without distorting in shape. The process involved utilising phenomenon of osmosis is explained as below -
Egg is placed in a mineral acid solution – after some time the egg is removed and placed in a hypertonic solution- size of the egg gradually decreases after some time and it shrivels due to o
New Question
10 months agoContributor-Level 10
We have,
At f(x) = 0.
x = 1 and x = -1.
At
At x = -1,
The maximum value of f(x)
Hence, option (D) is correct.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (iii)
SO2 is correct since photochemical fog is formed in the presence of sunlight in summers when NO and hydrocarbons are present in large amounts in the atmosphere. SO2 is not responsible for photochemical fog.
New Question
10 months agoContributor-Level 7
Students who are passionate about education and has a desire to work as teachers in K–12 institutions, colleges or universities can enroll in teaching courses. A bachelor's degree programme in education or their chosen field is usually the first step followed by certification or licensure as a teacher. Moreover, people who had previous jobs but want to change careers and get into teaching or education might sign up for teaching courses to acquire the appropriate credentials and training.
New Question
10 months agoContributor-Level 10
The latest cutoff information for the MBA course at Taxila Business School has not been released yet. However, according to the cutoff of the previous three academic years, the course and school show consistency. For better understanding, check the below table:
| Section | 2022 | 2023 | 2024 |
|---|---|---|---|
| Overall | 25 | 25 | 25 |
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
