Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 8
To check the availability of a PTE exam date, visit the official Pearson PTE website and choose your preferred exam according to your purpose of taking the exam. Once you choose your preferred exam, click on "check test availability and book".
From there on, you will be redirected to the "Find an Appointment" page. Enter your preferred nearby location in the search bar and you will get all the PTE test centres the nearest to your entered place. Click on the Calendar dropdown to see the available dates at these test centres; you can also check the available time slots on a given date by checking out the time slot given on the right
New Question
10 months agoContributor-Level 10
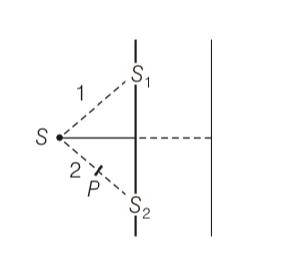
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation- when polariser is not used
A=Aperp+A
letA1= asinwt and A2=asin(wt+ )
now superposition principle for perpendicular polariser
AR= asinwt+ asin(wt+ )
AR=a(2cos sin(wt+ ))
AR=2acos sin(wt+ )
This eqn is also same for parallel polariser
AR=2acos sin(wt+ )
And we know that intensity is directly proportional to square of amplitude
(AR)2= (Aperp)2+(A)2
So resultant intensity is
I=4(a)2cos2 dt + 4(a)2cos2 dt
I= 8(a)2cos2 (1/2) &nb
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (i) & (iii)
Alkali metals and alkaline earth metals have low ionization enthalpy.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (ii) & (iii)
Both P and S have vacant d-orbital for which they execute extended covalency.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (i)
The electron gain enthalpy of 2p orbital is higher than that of the 3s orbital
New Question
10 months agoContributor-Level 10
IIT Delhi accepts the IIT JAM entrance exam, followed by counselling rounds for MSc admission across various specialisations. For Round 1 admission, the minimum and maximum ranks required for admission ranged from 9 to 513, respectively, for students in the General AI quota. Therefore, if you belong to the general category, you can certainly secure a seat at IIT Delhi. However, students must check which MSc specialisations are available with a cutoff rank of 100 and below, as cutoff ranks differ for various specialisations, ranks, and categories. Tabulated below is the IIT Delhi JAM first round cutoff 2025 for all MSc specialisations,
New Question
10 months agoNew Question
10 months agoContributor-Level 10
Somaiya School of Humanities and Social Sciences awards various scholarships to students as financial assistance. To be eligible for the financial aid, applicants must fulfill any one of the following criteria:
- EWS/BPL: Applicants with family income up to INR 8 LPA are eligible for financial Aid up to 100% concession on the available tuition fees.
- Defence Personnel: Wards of defence personnel deceased or disabled in the line of duty may receive a 50% concession on the available tuition fees.
- PWD: Applicants with benchmark disabilities (40% or even more) are eligible for a 50% concession on the available tuition fees.
New Question
10 months agoContributor-Level 10
For 2025 BTech admissions through UPTAC, key dates include registration, fee payment, and document upload from May 27 to June 30. Document verification and query response rounds are expected to take place between June and July, followed by choice filling, seat allotment, and confirmation rounds in August. Students should regularly check official updates to stay informed about changes in the UPTAC schedule.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
Yes, admissions for the BTech programme at Neelkanth Group of Institutions are currently open. Interested candidates who meet the eligibility criteria are encouraged to apply at the earliest. It is advisable to stay updated with official announcements regarding application deadlines and entrance exam requirements.
New Question
10 months agoContributor-Level 6
IIT Gandhinagar cutoff 2025 has been released for admission to MSc specialisations. The closing ranks in the round 1 were between the range of 338 and 396. The cutoff was released for MSc in Chemistry, MSc in Mathematics and MSc in Physics. The round 1 closing ranks for the above specialisations were 358, 396 and 338.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (iii) d-block
Lanthanum is the element with the atomic number 57 and it belongs to the d-block.
New Question
10 months agoContributor-Level 10
No, your school cannot deny you the MBSE 12th admit card even if you have skipped the pre-boards exam. If they still goes on to deny you the hall ticket you can straight away write it to the board asking them to issue your 12th hall ticket in duplicate as your school has failed to give you. Hall ticket for the MBSE 12th board exam is issued by the Mizoram Board of School Education (MBSE).
New Question
10 months agoContributor-Level 10
Students are advised to keep the MBSE 12th hall ticket till the release of the results. The MBSE 12th hall ticket contains the register number of the candidate. Thus student can easily check the MBSE 12th result using the register number.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (iii)
High amount of energy has to be supplied in order to overcome the e- - e - repulsion that arises when O- gets converted to O2- by accepting an electron
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (iv)
I+ possess the highest effective nuclear charge whereas I- possess the least among the given species.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Answer- (c, d)
Explanation- The simple Bohr model is not applicable to He4 atom because He4 has one more electron and electrons are not subject to central forces.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Answer- (b, d)
Explanation- when a radiation of energy fall on it some atoms would be excited but not all would be excite also no atom will go to 3 level some go to 2 level also but not all excite to 2 level. So by considering these facts we can say b and d option are correct
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (iii)
The lanthanoids which belong to the f-block elements are the elements where 4forbitals are filled progressively.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts