Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
Neelkanth Group of Institutions BTech course is available in various specialisations, including Computer Science and Engineering, Artificial Intelligence & Machine Learning, Artificial Intelligence & Data Science, Civil Engineering, Mechanical Engineering, and Electrical & Electronics Engineering. These specialisations are designed to align with current industry demands.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Explanation- According to Bohr model electrons having different energies belong to different levels having different values of n. So, their angular momenta will be different, as
L=nh/2 n
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Metallic character- Tendency to lose electrons.
Non-Metallic character- Tendency to accept electrons.
Across the period the metallic character decreases whereas non-metallic character increases across the period.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Explanation- If proton had a charge (+4/3)e and electron a charge (-3/4)e, then the Bohr formula for the H-atom remain same, since the Bohr formula involves only the product of the charges which remain constant for given values of charges.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Explanation- The transition of an electron from a higher energy to a lower energy level can appears in the form of electromagnetic radiation because electrons interact only electromagnetically.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Explanation- On removing one electron from He4 and He3, the energy levels, as worked out on the basis of Bohr model will be very close as both the nuclei are very heavy as compared to electron mass.Also after removing one electron from He4 and He3 atoms contain one electron and are hydrogen like atoms.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
(a) As the effective nuclear charge increases and shielding effect decreases across the periods thus the electronegativity increases on moving from left to right in the periodic table.
(b) As the number of shells increases down the group thus its ionisation enthalpy decreases down the group.
New Question
10 months agoContributor-Level 9
Ahmedabad Business School is a private college and is one of the prestigious colleges, located in Gujarat. It is also known as ABS and is approved by the All India Council for Technical Education (AICTE). ABS Gujarat courses are mainly offered to students in the field of Business Administration, Computer Applications, and Management.
New Question
10 months agoContributor-Level 7
Chandragupt Institute of Management accepts CAT/ XAT/ MAT for admission to MBA course. Students need to prepare for any one of the entrance exams and qualify as per the cutoff list for this college to be eligible for admissions for MBA at Chandragupt Institute of Management.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Explanation- the reason behind this is the binding energy or we can say the mass defect.
BE= mass defect (931MeV ). So whatever energy is liberated in fission and fusion changes the mass slighthly. That why there is a defect in mass
New Question
10 months agoContributor-Level 10
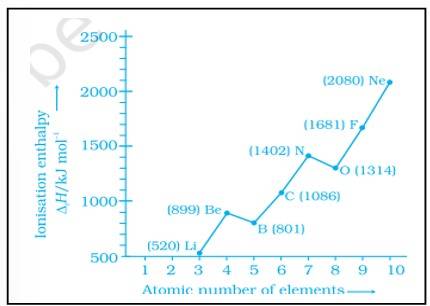
The deviation in the ionization enthalpy of some elements from the general trend can be explained by the points as given below:-
(i) The fully filled and half filled orbital provide extra stability due to the symmetry
(ii) The effective nuclear charge
(iii) The e- - e- repulsion which lead to instability
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation- when r
Let
F=
Where = (
Let p=
Electrostatic force is balanced by centripetal force
Mv2/r or v2=
New Question
10 months agoContributor-Level 7
To get admission to Chandragupt Institute of Management, students need to qualify for the basic eligibility criteria and relevant entrance exam. Find below the detailed admission criteria:
| Course | Eligibility Criteria | Selection Criteria |
|---|---|---|
| MBA | Graduation with a minimum 50% aggregate | CAT / XAT / MAT GD & PI |
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation- mp= 10-6
Mpc2=10-6
= 0.8
Wavelength associated with both of them is same
U(r)=-
Mvr=h
V=h/mr
Mv2/r=( ) ( )
13.6
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
(i) S
The ionization enthalpy increases across the period and decreases down the group. N possesses a half filled 2p orbital which provides it extra stability due to symmetry.
(ii)P
The non-metallic character increases across the period
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation- the energy of nth state En=-Z2R where R is constant and Z=24
The energy release ina transition from 2 to 1
The energy required to eject a n=4 electron is E4= Z2R1/16
So kinetic energy of auger electron is, KE= Z2R (3/4-1/16)=1/16Z2R
=
New Question
10 months agoContributor-Level 10
Yes, you can check your MBSE 12th result, if your MBSE 12th hall ticket is lost. You can check the MBSE 12th result from the school.
Hence, you need not worry, even if your MBSE 12th hall ticket is lost.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Exothermic reaction- The reaction in which heat is released
For example- CaO + CO2 → CaCO3 + Heat
Endothermic reaction- The reaction in which heat is absorbed.
For example- 2NH3 + heat → N2 +3H2
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation- in this type of situation centripetal force is balanced by electrostatic force
So mv2/r= -ke2/r2
According to bohr postuates
Mvr = h when n=1
On solving n
So after solving r= 0.51A0
So potential energy = -27.2eV , KE= mv2/2
=
But if R
If R>>r the electron moves inside the sphere with radius r’
Charge inside r’4=e
So r’ =
So r’4= 0.51A0R3
= 510(A0)4
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts