Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
Check below DESU placement highlights:
Particulars | Placement Statistics (2025) |
|---|---|
No. of Offers | 2300+ |
the highest Package | INR 11.5 LPA |
Average Package | INR 9 LPA |
No. of Companies participated | 165+ |
NOTE: The year for the data has not been mentioned, Hence considered to be for 2025
New Question
10 months agoContributor-Level 10
The key highlights of Sengunthar Engineering College BTech placements over the past three years are presented below:
Particulars | BTech Placement Statistics (2019) | BTech Placement Statistics (2020) | BTech Placement Statistics (2021) |
|---|---|---|---|
Median package | INR 2.4 LPA | INR 3.2 LPA | INR 3.2 LPA |
Total students | 130 | 136 | 112 |
Students placed | 112 | 115 | 96 |
Students selected for higher studies | 10 | 10 | 10 |
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(a, d) For house hold supplies, AC currents are used which are having zero average value over a cycle.
The line is having some resistance so power factor cos φ = R/Z≠0
so, φ not equal to π /2 ⇒ φ < π/2
i.e., phase lies between 0 and π /2.
New Question
10 months agoContributor-Level 10
Sengunthar Engineering College average package data is not released. However, as per the NIRF report 2022, the median package received over the past three years is presented below:
Particulars | Median Package (2019) | Median Package (2020) | Median Package (2021) |
|---|---|---|---|
BTech | INR 2.4 LPA | INR 3.2 LPA | INR 3.2 LPA |
MTech | INR 2.4 LPA | INR 2.4 LPA | INR 2.4 LPA |
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(c, d) When the AC voltage is applied to the capacitor, the plate connected to the positive terminal will be at higher potential and the plate connected to the negative terminal will be at lower potential.
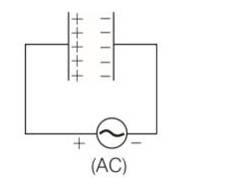
The plate with positive charge will be at higher potential and the plate with negative charge will be at lower potential. So, we can say that the charge is in phase with the applied voltage.
P= ErmsIrmscos
=90
So power, P = 0
New Question
10 months agoContributor-Level 10
DSEU has released its the highest package achieved in 2025 placement season. Check out the table below to know more:
Particulars | Placement Statistics (2025) |
|---|---|
the highest package | INR 11.5 LPA |
NOTE: The year for the data has not been mentioned, Hence considered to be for 2025.
New Question
10 months agoContributor-Level 10
The number of students placed over the past three years during Sengunthar Engineering College placements is presented below:
Particulars | BTech Placement Statistics (2019) | BTech Placement Statistics (2020) | BTech Placement Statistics (2021) |
|---|---|---|---|
Total students | 130 | 136 | 112 |
Students placed | 112 | 115 | 96 |
Students selected for higher studies | 10 | 10 | 10 |
Particulars | MTech Placement Statistics (2019) | MTech Placement Statistics (2020) | MTech Placement Statistics (2021) |
Total students | 1 | 10 | 10 |
Students placed | 1 | 10 | 10 |
New Question
10 months agoContributor-Level 10
The placement trend recorded at Sengunthar Engineering College over the past three years is presented below:
Particulars | BTech Placement Statistics (2019) | BTech Placement Statistics (2020) | BTech Placement Statistics (2021) |
|---|---|---|---|
Median package | INR 2.4 LPA | INR 3.2 LPA | INR 3.2 LPA |
Total students | 130 | 136 | 112 |
Students placed | 112 | 115 | 96 |
Students selected for higher studies | 10 | 10 | 10 |
Particulars | MTech Placement Statistics (2019) | MTech Placement Statistics (2020) | MTech Placement Statistics (2021) |
Median package | INR 2.4 LPA | INR 2.4 LPA | INR 2.4 LPA |
Total students | 1 | 10 | 10 |
Students placed | 1 | 10 | 10 |
New Question
10 months ago
Contributor-Level 10
Praxis Tech School Bangalore has a good record of placements for its PGP Data Science & AI Skill course. The highest package offered during Praxis Tech School Bangalore placements 2023 stood at INR 23 LPA. Check out the key highlights of Praxis Tech School Bangalore placements the Class of 2023 in the table below:
Particulars | Placement Statistics (2023) |
|---|---|
the highest package | INR 23 LPA |
Average package | INR 13.50 LPA |
Median package | INR 13 LPA |
Total recruiters | 80+ |
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Transition elements are named so because they form a bridge between s-block elements and p-block elements. Zn, Cd and Hg are among those elements that are dblock elements but they do not exhibit most of the properties of transition elements.
New Question
10 months agoContributor-Level 10
The key highlights of Sengunthar Engineering College placements in recent past are presented below:
Particulars | BTech Placement Statistics (2021) | MTech Placement Statistics (2021) |
|---|---|---|
Median package | INR 3.2 LPA | INR 2.4 LPA |
Total students | 112 | 10 |
Students placed | 96 | 10 |
Students selected for higher studies | 10 | 0 |
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
According to the question power transferred Is P = I2 Z cos?
as we know cos? = R/Z
R>0 and Z>0
cos? >0 so P>0
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
As the atomic size of F is much smaller which leads to high e- - e - repulsion upon addition of electrons thus its electron gain enthalpy is less than that of Cl.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
We have to transmit energy (power) over large distances at high alternating voltages, so current flowing through the wires will below because for a given power (P).
P= ErmsIrms, Irms is low when Erms is high.
Power loss= I 2 rms R= low
New Question
10 months agoContributor-Level 10
Check below placement statistics recorded during DSEU placements 2025:
Particulars | Placement Statistics (2025) |
|---|---|
No. of Offers | 2300+ |
the highest Package | INR 11.5 LPA |
Average Package | INR 9 LPA |
No. of Companies participated | 165+ |
NOTE: The year for the data has not been mentioned, hence considered for 2024
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: As we move across the periodic table the ionization energy increases because of the increase in the effective nuclear charge and decrease of the shielding effect as more and more electrons get added in the same orbital.
Thus group 1 has lower ionization enthalpy compared to that of group 17 and also group 1 by losing one electron it will acquire the nearest noble gas electronic configuration which also contributes towards its lower ionization enthalpy.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(c, d) According to the question, the current increases on increasing the frequency of supply. Hence, the reactance of the circuit must be decreases as increasing frequency.
For a capacitive circuit, capacitive reactance Xc=
For an CR circuit Z= when frequency increases X decreases.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
The following points make long form of periodic table better than Mendeleev's periodic table :-
(a) It is a periodic function of atomic number
(b) Elements are grouped as per there outermost electronic configuration
(c) Proper segregation of metals and non-metals
(d) More appropriate position of group VII.
New Question
10 months agoContributor-Level 10
Tritya Institute of Event Management selects students based on aptitude test or entrance exam, depending on the course. If two candidates have the same TIEMAT/CMAT/CAT/MAT score, preference will be given to the one who completes the admission process first.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Mendeleev's arranged the elements as the periodicity of their atomic weights.
The drawbacks of Mendeleev's periodic table are as follows:-
(a) The position of hydrogen in the periodic table is not specified
(b) Isotopes are not included in the periodic table
(c) Elements with higher atomic mass are placed before the elements with lower atomic mass. For e.g- Co & Ni
(d) Gaps are left in his table considering the fact that more elements are yet to be discovered
(e) Inappropriate position of group VII.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
