Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoNew Question
11 months agoContributor-Level 10
The fee for courses offered by NMIMS School of Commerce, Dhule are as follows:
- BBA: INR 4.4 lakh
- BCom: INR 2.9 lakh
- BBA LLB: INR 5.1 lakh
Candidates are required to visit the official website of the institute to fill out the application form to participate in the admission process. Candidates who clear the admisison process are eligible to pay the course fee and appear fro the regular classes.
New Question
11 months agoContributor-Level 10
13.14 Let the atomic mass of a substance be = M and the density of the substance be =
Avogadro's number, N = 6.023
Volume of N number of molecules = ……. (i)
Volume of one mole of a substance = …… (ii)
Equating (i) and (ii), we get

New Question
11 months agoContributor-Level 10
The placement rate recorded during Ellenki College of Engineering and Technology placements was 95%. Check out the table below for branch wise placement rate during 2024 placement season:
Branch | Placement percentage |
|---|---|
CSE | 95% |
CSE - AI & ML | 95% |
CSE – Data Science | 95% |
CSE – Cyber Security | 90% |
ECE | 75% |
EEE | 75% |
CIVIL | 75% |
MECH | 75% |
MBA | 95% |
New Question
11 months agoContributor-Level 10
CII Institute of Hospitality - ITC Maurya is a premier institution for students to achieving high-quality Hospitality education. The UGC-recognized courses offered by CIIIH ITC Maurya include the BTS (Bachelor in Tourism Studies), as well as global-standard Swiss Hospitality courses sponsored by Ecole Hôtelière de Lausanne (EHL).
CIIIH ITC Maurya provides 3 distinct specializations in its EHL Diploma programs, serving different departments of the Hospitality industry. The infrastructure includes world-class amenities at the ITC Maurya hotel, and the facilities provided by the Learning Management System (LMS) by EHL. The leadin
New Question
11 months agoContributor-Level 10
The Ramrao Adik Institute of Technology offers an M.Tech programme at the postgraduate level. For admission, students are required to submit an application form. The fee to fill out and submit the form is as follows:
Course | Application Fees 2025 |
|---|---|
BTech | INR 2,150 |
Note: The below-mentioned fee is as per official sources. However, it is subject to change.
New Question
11 months agoContributor-Level 10
The highest package offered during Ellenki College of Engineering and Technology placements 2024 stood at INR 20 LPA. The table below presents the same:
Particulars | Placement Statistics (2024) |
|---|---|
the highest package | INR 20 LPA |
New Question
11 months agoContributor-Level 10
13.13 According to law of atmospheres, we have
n2 = n1 exp [ -mg (h2 – h1)/ kBT] ….(i)
where is the number of density at height and is the number of density at height
mg is the weight of the particle suspended in the gas column
Density of the medium =
Density of the suspended particle =
Mass of one suspended particle = m'
Mass of medium displaced = m
Volume of the suspended particle = V
According to Archimedes's principle for a particle suspended in a liquid column, the effective weight of the suspended particle is given as:
Weight of the medium displaced – weight of the suspended particle = mg – m'g
= mg- V = mg –
New Question
11 months agoNew Question
11 months agoContributor-Level 10
Yes, while applying for M.Tech admissions at Ramrao Adik Institute of Technology, students are required to upload scanned copies of various academic and personal documents. Check the below list to know the required documents:
- Class 10 marksheet
- Class 12 marksheet
- Migration certificate
- School leaving certificate/transfer certificate
- Category certificate (if applicable)
- UG marksheets
- UG degree
- Income certificate (applicable for candidates seeking admission via EWS category)
- Domicile certificate (if applicable)
New Question
11 months agoContributor-Level 10
13.12 Rate of diffusion of hydrogen, = 28.7 cm3 s–1
Rate of diffusion of another gas, = 7.2 cm3 s–1
According to Graham's law of diffusion, we have:
= , where = molecular mass of hydrogen = 2.02 g and is the molecular mass of the unknown gas
= 2.02= 32.09 = Molecular mass of Oxygen
Hence, the unknown gas is Oxygen.
New Question
11 months agoContributor-Level 10
The placements for 2024 at Ellenki College of Engineering and Technology are still ongoing. As of now 43 students have been placed. The following table presents the placement statistics for Ellenki College of Engineering and Technology recorded in 2024
Particulars | Placements Statistics (2024) |
|---|---|
the highest package | INR 20 LPA |
Average package | INR 3.5 LPA |
Number of Students Placed | 226 |
Number of Offers | 250 |
Companies Participated | 50+ |
Placement Percentage | 95% |
Check out the branch wise placement package in the table below:
Branch | Number of Students Placed | the highest Package |
|---|---|---|
CSE | 150 | INR 15 LPA |
CSE-AI & ML | 8 | INR 7 LPA |
CSE- Data Science | 10 | INR 6.5 LPA |
CSE – Cyber Security | 7 | INR 7 LPA |
ECE | 15 | INR 7 LPA |
EEE | 3 | INR 7.5 LPA |
MECH | 10 | INR 5 LPA |
Civil | 8 | INR 5 LPA |
MBA | 15 | INR 7 LPA |
New Question
11 months agoContributor-Level 10
13.11 Length of the narrow bore, L = 1 m = 100 cm
Length of the mercury thread, l = 76 cm
Length of the air column between mercury and the closed end, = 15 cm
Since the bore is held vertically in air with the open end at the bottom, the mercury length that occupies the air space is 100 – (76 + 15) = 9 cm
Hence, total length of the air column = 15 + 9 = 24 cm
Let h cm of mercury flow out as a result of atmospheric pressure.
Length of the air column in the bore = 24 + h cm
Length of the mercury column = 76 – h cm
Initial pressure, = 76 cm of mercury
Initial volume, = 15
Final pressure, = 76 – (76 – h) = h cm
New Question
11 months agoNew Question
11 months agoContributor-Level 10
13.10 Pressure inside the cylinder containing nitrogen, P = 2.0 atm = 2 Pa
Temperature inside the cylinder, T = 17
Radius of nitrogen molecule, r = 1.0 Å = 1 m
Diameter of nitrogen molecule, d = 2 m
Molecular mass of nitrogen molecule, M = 28 u = 28 g (assume) = 28 kg
The root means square speed of nitrogen is given by the relation

R is the universal gas constant = 8.314 J/mole/K
Hence
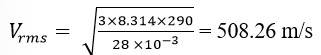
The mean free path is given by
where k = Boltzmann constant = 1.38 kg-
= 1.11 m
Collision frequency = = = 4.57 /s
Collision time, T = =S= 3.93 s
T
New Question
11 months agoContributor-Level 10
Yes, the application forms for the preferred RAIT M.Tech course can be filled online. Students who wish to apply online can do so via DY Patil Navi Mumbai's online admission portal. Check below for the steps to apply:
1. Go to the official website of the institute.
2. Fill out the registration form appearing on the screen.
3. Log in to the account with the credentials created in the earlier step.
4. Upload the required documents and pay the application fees of INR 2,150.
5. Preview and submit the form.
New Question
11 months agoContributor-Level 10
13.9 Temperature of the helium atom, = – 20 °C = 253 K and temperature of argon atom be =
Atomic mass of helium, = 4.0 u
Atomic mass of Argon, = 39.9 u
Let be the rms speed of Argon and be the rms speed of Helium
From the relation of we get
rms speed of Argon,
rms speed of Helium,
Since both the speeds are equal, we get
= or = or = = = 2523.675 K = 2.523 K
New Question
11 months agoContributor-Level 10
Diffraction leads to the formation of patterns of varying intensity. When around obstacles, waves bend and spread through the narrow opening, it is called diffraction. The interference results in a new wave pattern and involves the superposition of two or more coherent waves. Both these phenomena produce patterns of light and dark regions; the interference results from the combination of multiple waves and the diffraction arises from a single wave interacting with an aperture or obstacle. When the size of the aperture or obstacle is comparable to the wavelength of the wave, diffraction patterns are typically observed.
New Question
11 months agoContributor-Level 10
13.8 (a) According to Avogadro's law, the three vessels will contain an equal number of the respective molecules. This number is equal to Avogadro's number, N = 6.023
(b) The root mean square speed ( of a gas of mass m and temperature T is given by the relation . Where k is Boltzmann constant. For the given gases, k and T are constants. Hence depends only on the mass of the atoms ![]()
Therefore, the root mean square speed of the molecules in the three cases is not the same. Among Neon, Chlorine and Uranium hexafluoride, the mass of the neon is the smallest, so Neon will have the highest root mean square sp
New Question
11 months agoContributor-Level 10
St Anne's Degree College for Women BCA admission starts when Class 12 results are announced. Once candidates have secured a valid score in their Class 12 exams they will have to apply for admission through the college website. Candidates applying BCA admission should make sure they are eligible for admission, the eligibility criteria to secure admission in the programme is a passing grade in Class 12.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
