Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
3.27. (a) Element belonging to nitrogen family (group 15) e.g., nitrogen.
(b) Element belonging to alkaline earth family (group 2) e.g., magnesium.
(c) Element belonging to oxygen family (group 16) e.g., oxygen.
New Question
11 months agoContributor-Level 10
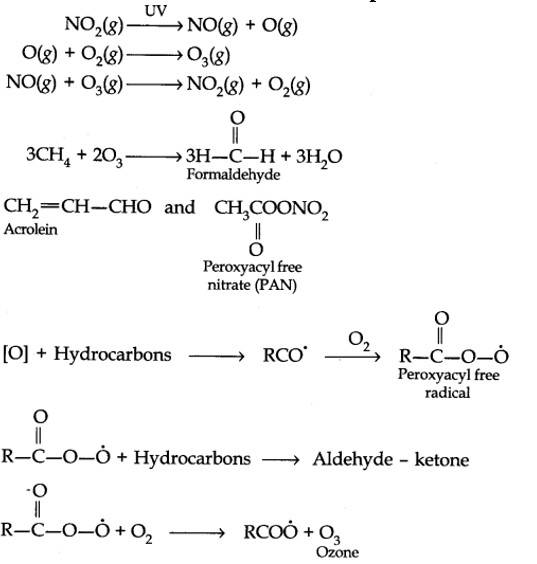
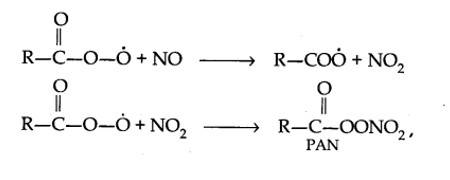
The word smog is a combination of smoke and fog. It is a type of air pollution that occurs in many cities throughout the world. Classical smog occurs in a cool, humid climate. It is also called reducing smog. Whereas photochemical smog (photo means light) occurs in warm and dry sunny climates. It has a high concentration of oxidising agents and therefore, it is also called oxidising smog.
New Question
11 months agoContributor-Level 10
This is mainly due to the large number of industries and power plants in the nearby areas. Acid rain has vapours of sulphuric acid dissolved in it. When it comes in contact with various statues or monuments, the acid reacts chemically with marble (calcium carbonate).
CaCO3 + H2SO4 à CaSO4 + H2O + CO2
As a result, the monument is being slowly disfigured, and the marble is getting discoloured and lustreless.
New Question
11 months agoContributor-Level 10
1.24 Ferromagnetic substances would make better permanent magnets because when the ferromagnetic substance is placed in a magnetic field, all domains get oriented in the direction of magnetic field and strong a magnetic effect is produced.
New Question
11 months agoContributor-Level 10
CO2 is mainly responsible for the greenhouse effect. Other greenhouse gases are methane, nitrous oxide, water vapours, CFCs (chlorofluorocarbons) and Ozone.
New Question
11 months agoContributor-Level 10
3.26.
Metals | Non-metals |
1. They have strong tendency to lose electrons to form cations. | 1. They have strong tendency to accept electrons to form anions. |
2. Metals are strong reducing agents. | 2. They are strong oxidising agents. |
3. Metals have low ionization enthalpies. | 3. Non-metals have high ionization enthalpies. |
4. They form basic oxides and ionic compounds. | 4. They form acidic oxides and covalent compounds. |
New Question
11 months agoContributor-Level 10
Carbon monoxide binds to haemoglobin to form carboxyhaemoglobin, which is about 300 times more stable than the oxygen-haemoglobin complex. In blood, when the concentration of carboxyhaemoglobin reaches about 3–4 per cent, the oxygen-carrying capacity of blood is greatly reduced. This oxygen deficiency results into headache, weak eyesight, nervousness and cardiovascular disorder. This is the reason why people are advised not to smoke. In pregnant women who have the habit of smoking the increased CO level in blood may induce premature birth, spontaneous abortions and deformed babies. On the other hand, CO2 does not combine with ha
New Question
11 months agoContributor-Level 10
1.23 An n-type semiconductor conducts because of the presence of extra electrons. Therefore, a group 14 element can be converted to n-type semiconductor by doping it with a group 15 element.
New Question
11 months agoContributor-Level 10
Tropospheric pollution occurs due to the presence of undesirable solid or gaseous particles in the air. The following are the major gaseous and particulate pollutants present in the troposphere:
1. Gaseous air pollutants: These are oxides of sulphur, nitrogen and carbon, hydrogen sulphide, hydrocarbons, ozone and other oxidants.
2. Particulate pollutants: These are dust, mist, fumes, smoke, smog etc.
New Question
11 months agoContributor-Level 10
1.22 The colour develops because of the presence of electrons in the anionic sites. These electrons absorb energy from the visible part of radiation and get excited. For example, when crystals of NaCl are heated in an atmosphere of sodium vapours, the sodium atoms get deposited on the surface of the crystal and the chloride ions from the crystal diffuse to the surface to form NaCl with the deposited Na atoms. During this process, the Na atoms on the surface lose electrons to form Na+ ions and the released electrons diffuse into the crystal to occupy the vacant anionic sites. These electrons get excited by absorbing energy from the visi
New Question
11 months agoContributor-Level 10
Environmental chemistry is a branch of environmental studies. Environmental studies deal with the sum of all social, economical, biological, physical and chemical interrelations with our surroundings. Environmental chemistry deals with the study of the origin, transport, reactions, effects and fates of chemical species in the environment
New Question
11 months agoContributor-Level 7
Biotech is a industry that holds $150 billion of evaluation and aims to surpass $300 billon over the time of 2030.
The career can be build as a R&D Scientiest, Genetic engineer, Bio process Engineer etc.
Avg Salary starts from 4-6.5 LPA For B.Tech in Biotechnology.
Top colleges are :
- Banaras Hindu university
- Institute of Chemical Technology
- Presidency University, Kolkata
- Lovely professional university
New Question
11 months agoContributor-Level 10
3.25. Isotopes have same number of electrons and protons, only the number of neutrons is different. The atomic number remains the same and only atomic mass differs. Hence the ionization energy of the isotopes of a chemical element remains the same.
New Question
11 months agoContributor-Level 10
The top recruiting companies that hire students from various Finance colleges in India are listed in the table below:
College Name | Top Recruiters |
|---|---|
BCG, Accenture Strategy, Deloitte, etc. | |
Microsoft, Google, EY, HCL, Deloitte, Accenture, Adani Group, Airtel, BCG, Capgemini, TCS, PwC, etc. | |
Accenture, Amazon, Microsoft, Deloitte, TCS, HCL, Cognizant, etc. | |
Accenture, Google, ICRA, Deloitte, Bajaj, KPMG, etc. | |
Adani Wilmar, Bajaj Auto, Zomato, and Praxis Global Alliance, etc. |
Disclaimer: This information is sourced from the official website and may vary.
New Question
11 months agoContributor-Level 10
The average placement salary of a Finance graduate varies depending on skills, knowledge, academic background, college reputation, etc. However, the following table provides an average placement salary for students who have completed Finance courses from some of the top colleges in India:
College Name | Average Package |
|---|---|
INR 22.8- 30 LPA | |
INR 30 LPA | |
INR 27 LPA | |
INR 21- 26.4 LPA | |
INR 26.04 LPA |
Disclaimer: This information is sourced from the official website and may vary.
New Question
11 months agoBeginner-Level 5
Vidya PratishthanVidya Pratishthan is a charitable trust in Baramati, Maharashtra, established in 1972 to provide affordable quality educationinstitutions across 260 acres, offering education from kindergarten to doctoral levels. The trust was founded by Hon. Shri. Sharadchandraji Pawar.
Here's a more detailed look:
Founding and Purpose:
Vidya Pratishthan was established to provide accessible and quality education to students in Baramati and surrounding areas.
Scale and Scope:
The trust manages a large campus with 29 institutions, covering a wide range of educational levels.
VPKBIET:
Vidya Pratishthan's Kamalnayan Bajaj Inst
New Question
11 months agoContributor-Level 10
Students can calculate the ROI of studying a Finance degree at various colleges in India, by using the data provided below:
College Name | Average Fees | Average Package |
|---|---|---|
INR 14.5 Lakh- 24 Lakh | INR 22.8- 30 LPA | |
INR 4.1 Lakh- 27 Lakh | INR 30 LPA | |
INR 3.5 Lakh- 20.5 Lakh | INR 27 LPA | |
INR 6.01 Lakh | INR 21- 26.4 LPA | |
INR 6.93 Lakh- 24.2 Lakh | INR 26.04 LPA |
Disclaimer: This information is sourced from the official website and may vary.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
Yes, there are 190+ government Finance colleges in India. Students can take a look at the table below to know the course fees at these top Finance colleges in India:
College Name | Average Fees |
|---|---|
INR 1.2 Lakh- 1.35 Lakh | |
INR 3.5 Lakh- 20.5 Lakh | |
INR 4.1 Lakh- 27 Lakh | |
INR 12 Lakh | |
INR 2.2 Lakh- 2.98 Lakh |
Disclaimer: This information is sourced from the official website and may vary.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts