Ask & Answer: India's Largest Education Community
All Questions
New Question
9 months agoContributor-Level 10
Yes, joining the best Perfusion Technology colleges in India can be worthwhile for you. For that, you need to check its ROI. Listed below are some colleges along with their tuition fees and average placement package:
Top Colleges | Tuition Fee | Average Placement Package |
|---|---|---|
JIPMER Puducherry Courses | INR 4,800 | INR 8 LPA |
Sree Chitra Tirunal Institute for Medical Sciences and Technology Courses | INR 10,000 | INR 5.04 LPA |
Sri Ramachandra Institute of Higher Education and Research Courses | INR 3 lakh | INR 5.25 LPA |
JSS Medical College Courses | INR 5.4 lakh | INR 9.75 LPA |
Medical College, Bharati Vidyapeeth Courses | INR 3 lakh | INR 4.4 LPA |
Disclaimer: This information is sourced from official website/ media reports/ NIRF website and may vary.
New Question
9 months agoContributor-Level 10
Chlorine nitrate as hydrolysis gives hypohalous acid and nitric acid as,
Chlorine nitrate on reaction with HCl produces Cl2 and HNO3 as,
New Question
9 months agoContributor-Level 10
Yes, there are about 8 top Perfusion Technology colleges in India that offer Diploma courses to their students:
Top Colleges | Tuition Fee |
|---|---|
Sree Chitra Tirunal Institute for Medical Sciences and Technology | INR 10,000 |
Institute of Post Graduate Medical Education and Research | INR 30,000 |
Apollo Institute of Hospital Management and Allied Sciences | INR 8,000 |
SVMC - Sri Venkateswara Medical College, Tirupati | INR 35,400 |
Disclaimer: This information is sourced from official website and may vary.
New Question
9 months agoContributor-Level 10
Students at Sheridan can choose from more than 140+ options if they're looking for a UG or PG degree. Apart from Business Management, IT, and Software Engineering, some other popular courses at the institute include:
- Design
- Law
- Mass Communication and Media
- Humanities
- Arts
- Science
- Architecture
- Accounting
- Medical Science
- Education
New Question
9 months agoContributor-Level 10
Yes, there are many affordable Perfusion Technology colleges in India. Some of them are mentioned below along with their tuition fees:
Low-cost Colleges | Tuition Fee |
|---|---|
JIPMER Puducherry | INR 4,800 |
AIIMS Rishikesh | INR 650 |
Sree Chitra Tirunal Institute for Medical Sciences and Technology | INR 10,000 |
Institute of Post Graduate Medical Education and Research | INR 30,000 |
St. John's National Academy of Health Sciences | INR 97,200 |
Disclaimer: This information is sourced from official website and may vary.
New Question
9 months agoContributor-Level 10
As per popularity basis, listed below are the top Perfusion Technology colleges in India along with their tuition fees:
Top Colleges | Tuition Fee |
|---|---|
JIPMER Puducherry | INR 4,800 |
AIIMS Rishikesh | INR 650 – INR 1.11 lakh |
Sanjay Gandhi Postgraduate Institute of Medical Sciences | INR 2.2 lakh |
Sree Chitra Tirunal Institute for Medical Sciences and Technology | INR 10,000 |
Institute of Post Graduate Medical Education and Research | INR 30,000 |
Disclaimer: This information is sourced from official website and may vary.
New Question
9 months agoContributor-Level 10
About 80+ best Perfusion Technology colleges are there in India. Some of the top Perfusion Technology colleges in India include Postgraduate Institute of Medical Education and Research, JIPMER Puducherry, AIIMS Rishikesh, Sanjay Gandhi Postgraduate Institute of Medical Sciences, All India Institute of Medical Sciences, Bhubaneswar, Sree Chitra Tirunal Institute for Medical Sciences and Technology, etc.
Of these, 57 colleges are privately owned and 17 colleges are owned by public/government organisations. Students can secure admission on merit basis or through entrance exam like KCET, CUET, etc.
New Question
9 months agoContributor-Level 10
The admission process for Moople Institute Barrackpre comprises of several steps starting from application/ registration followed by selection and final admission confirmation. Students need to meet the basic admission criteria set by the institute to be eligible for admission. Those who qualify for the desired course may follow the steps listed below to complete the institute;s admission process:
Step 1: Application Submission - Prospective students must fill out and submit the application form available on the institute's official website.
Step 2: Admission Counselling - After submitting the application form, an admissio
New Question
9 months agoContributor-Level 10
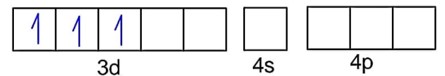
A.
Fe3+ - 3d54s0
F- is weak field ligand, so no pairing of electrons :
Number of unpaired electrons, n = 5
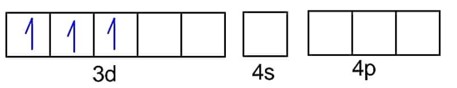
B.
CN- is strong field ligand, so pairing of electrons takes place.
Number of unpaired electrons, n = 1
C.
New Question
9 months agoContributor-Level 10
Electronic configuration of the given ions is :
Hence, Eu2+ and Tb4+ have half | filled f-orbitals and Yb2+ have completely filled f-orbitals.
New Question
9 months agoNew Question
9 months agoContributor-Level 10
In 3d-series, all metals except Cu have negative value of
New Question
9 months agoContributor-Level 10
Fluorine forms only one oxoacid which is hypofluorous acid HOF because it shows only – 1 oxidation state. Which is due to its the smallest size among halogens & the highest electronegativity.
New Question
9 months agoNew Question
9 months agoContributor-Level 10
Higher the E.N. difference between hydrogen and other atom then higher be the strength of intermolecular H-bond
Here, order of difference in E.N is
O - H > N – H > C - H
Hence, correct order of H bond strength is,
CH4 < HCN < NH3
New Question
9 months agoContributor-Level 10
Leaching involves the given reaction,
Here, O2 is required for formation of Au (l) cyanide complex but no complex in absence of O2.
In above displacement reaction, Zn is oxidized.
New Question
9 months agoContributor-Level 10
Structure PCl5 is trigonal bipyramidal,
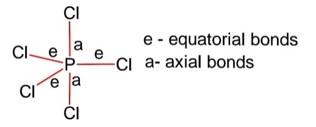
Hybridization of P is sp3d
Equatorial bonds lie in a plane
Axial bonds are longer than equatorial bonds so axial bonds are weaker than equatorial bonds.
New Question
9 months agoContributor-Level 10
Fluorine forms only one oxoacid which is hypofluorous acid HOF because it shows only – 1 oxidation state. Which is due to its the smallest size among halogens & the highest electronegativity.
New Question
9 months agoContributor-Level 10
Higher the E.N. difference between hydrogen and other atom then higher be the strength of intermolecular H-bond
Here, order of difference in E.N is
O - H > N – H > C - H
Hence, correct order of H bond strength is,
CH4 < HCN < NH3
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts