Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 8
In order to prepare for the KIITEE Law exam, a number of books covering various areas are suggested. "Quantitative Aptitude by R.S. Aggarwal" is a well-liked option for quantitative aptitude. For English, "Objective General English by S.P. Bakshi" or "High School English Grammar & Composition Revised Edition" by Wren and Martin are recommended. It is advised to use "A Modern Approach To Verbal and Non-Verbal Reasoning" by R.S. Aggarwal or "A New Approach to Reasoning Verbal and Non-Verbal" by B.S. Sijwali and Indu Sijwali for reasoning. Gaurav Mehta's "Universal's Guide to LL.M. Entrance Examination" is also re
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Option (C) The combustion reaction of methane is given below
CH4 + O2 -> CO2 + H2O
In this reaction, water and carbon dioxide are formed.
According to the reaction, 1 mole of methane gives 2 moles of water. So, 16 g of methane gives 36 g of water
Thus, X is false but R is true
New Question
10 months agoContributor-Level 10
When selecting DIT University for BSc, students should consider factors such as specialisations offered, curriculum relevance, faculty expertise, and placement support. It's also important to assess industry exposure, internship opportunities, tuition fees, infrastructure, and student feedback to ensure that the programme aligns with long-term career goals.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Option (C)
In the decimal portion, only trailing or final zeros are significant. This is in accordance with the rules of significant figures.
The given number, 0.200 has 3 significant figures whereas the number, 200 has only one significant figure because zeros present on the right side or at the end of the decimal number are significant and it is provided that they are present on the right side of the decimal point.
Also, zeros are not significant in numbers without decimals. Thus, X is true but R is false.
New Question
10 months agoContributor-Level 7
Yes, admissions are currently ongoing for admission to United School of Business Management. Students can apply directly on the college's official website to fill the application form for UG and PG courses.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Option (B)
Atomic mass unit is defined as a mass that is equal to accurately the mass of carbon-12 atom.
When scientists compare the relative atomic masses of the elements to the mass of carbon, they find that they are near to a whole number value.
So, both X and B are true but R is the correct explanation of A.
New Question
10 months agoContributor-Level 10
Parul University and New L.J. Institute are both good for B.Tech, but they have different strengths. Parul has a bigger campus, more diverse courses, and decent placement opportunities, especially in tech branches. New L.J. Institute is more focused, especially in CSE, with good labs and academic discipline.
New Question
10 months agoContributor-Level 7
United School of Business Management applications are accepted in online mode. The application fee for all the UG and PG courses is INR 500. Students can follow steps given below to apply for United School of Business Management admissions:
- Visit the official website at USBM.
- Fill the application form with details around personal and academic details.
- Complete the form and pay the application fee.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
(i) Both A and R are true and R is the correct explanation of A.
Explanation: The molecular formula gives the actual number of different atoms present in a molecule and the empirical formula gives a simple whole number ratio of different atoms present in a molecule.
Since ethene C2H4 can be further simplified, the empirical formula becomes CH2. The molecular mass of C2H4 is 28 g/mol and the empirical mass of CH2 is 14 g/mol.
Hence, the empirical mass of ethene is half of its molecular mass.
New Question
10 months agoContributor-Level 6
At JIMS Engineering Management Technical Campus, students can get admission into various UG courses such as BE/BTech, BCom, BCA, BBA and BA LLB. The institute offers admission to these courses based on several national and college level exams. Candidates can find more information on courses offered at the institute on this page.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
PSNA College of Engineering and Technology has released its placement details on its official website. Refer to the table below for more placement information for placements held in 2023-2025:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
the highest Package | INR 15.14 LPA | INR 18 LPA | INR 44 LPA |
Average Package | INR 3.6 LPA | INR 4 LPA | INR 4.2 LPA |
Total Recruiters | 114 | 103 | 104 |
Total Offers | 1401 | 1010 | 1107+ |
Students Placed | 1116 | 1139 | 1039 |
New Question
10 months agoContributor-Level 10
PSNA College of Engineering and Technology has released its placement brochure. Annually, leading companies take part in placing graduating students. For more information on key takeaways for the PSNA College of Engineering and Technology placements, refer to the table below:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
the highest Package | INR 15.14 LPA | INR 18 LPA | INR 44 LPA |
Average Package | INR 3.6 LPA | INR 4 LPA | INR 4.2 LPA |
Total Recruiters | 114 | 103 | 104 |
Total Offers | 1401 | 1010 | 1107+ |
Students Placed | 1116 | 1139 | 1039 |
New Question
10 months agoContributor-Level 10
The PSNA College of Engineering and Technology is well known for the excellent placement packages provided to its students. For more information on placement packages and recruiters for the PSNA College of Engineering and Technology placements 2023-2025, refer to the table below:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
the highest Package | INR 15.14 LPA | INR 18 LPA | INR 44 LPA |
Average Package | INR 3.6 LPA | INR 4 LPA | INR 4.2 LPA |
Total Recruiters | 114 | 103 | 104 |
Total Offers | 1401 | 1010 | 1107+ |
Students Placed | 1116 | 1139 | 1039 |
New Question
10 months agoContributor-Level 10
Yes, candidates can apply for MBA course with a bachelor's degree at JD Institute of Fashion Technology Bangalore. The eligibility criteria for this course is based on merit. Through its dynamic teaching approach, the MBA in Fashion Design from JD Institute programme equips students with the expertise and aptitude required to run and manage businesses effectively across fashion and lifestyle sectors.
New Question
10 months agoContributor-Level 10
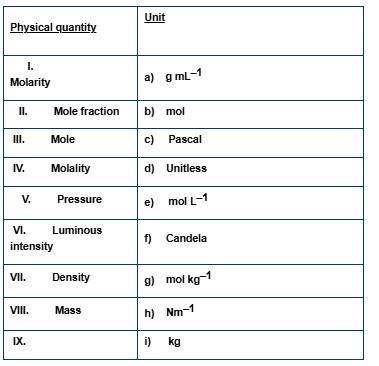
This is a Matching Type Questions as classified in NCERT Exemplar
(i) → (e) (ii) → (d) (iii) → (b) (iv) → (g) (v) → (c), (h) (vi) → (f) (vii) → (a) (viii) → (i)
Explanation:
(i) It is defined as the number of moles of solute dissolved in 1 litre of solution. So, the unit of molarity is mol L-1
(ii) Mole fraction is defined as the number of moles of a constituent divided by the total number of moles. So, it is unitless.
(iii) Mole is the unit to measure the amount of an atom or molecule in SI system. it is symbolized as "mol".
(iv) It is defined as the number of moles of solute dissolved in 1 kg of
New Question
10 months agoContributor-Level 10
CUET, UPCATET and ICAR AIEEA are the main entrance exams you can aim to pursue a BSc Home Science course. Some colleges also consider merit-based admissions; on the other hand some may require specific subjects at the 10+2 level, such as Physics, Chemistry, and Biology.
CUET (UG): A national-level entrance exam conducted by NTA (National Testing Agency) and is accepted by many colleges across the country for B.Sc. Home Science admissions.
ICAR AIEEA: The Indian Council of Agricultural Research All India Entrance Examination is another popular entrance exam that offers Home Sciencec courses.
UPCATET : Uttar Pradesh Common Admission Test
New Question
10 months agoContributor-Level 10
A total of 104 recruiters are seen during the PSNA College of Engineering and Technology placements in 2025. Check out the table below to know more about PSNA College of Engineering and Technology placements recruiters for 2023-2025:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
Total Recruiters | 114 | 103 | 104 |
New Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
(i)- (b); (ii) - (c); (iii) - (a); (iv) - (e); (v) - (d)
(i) 88 g of CO2
The number of moles is given by the following formula,
Moles = - (1)
So, the number of moles of CO2 is calculated by using equation (1) as follows
Moles of CO2 = = 2 mol
Thus, option (i) from column I is matched with (b) from column II
(ii) 1 mol of H2O gives 6.022*1023 molecules. So, 6.022*1023 molecules contain 1 mol of H2O . Thus, option (ii) from column I is matched with (b) from column II
(iii) 5.6 liters of O2 at STP
1 mol of gas occupies 22.4 liters of O2 . For 5.6 lit
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (A), (D)
Law of conservation of mass: According to this law, the mass is neither created nor destroyed during a chemical reaction in an isolated system. It can only be transformed from one form to another. Burning of wood is an example of conservation of mass. Law of definite proportion:
According to this law, a compound contains exactly the same elements in the fixed proportion by mass. For example, pure water consists of 11.196 H and 88.9% o by mass.
Law of multiple proportion: According to the law of multiple proportion, when two elements react to form two
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts