Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 6
Check out tips to start GMAT preparation:
- Take a mock test.
- It points out your weak spots in exam sections. This will help you build a smart study plan that focuses your time.
- Consistent work brings results.
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
Sodium reacts with Dihydrogen to form sodium hydride which is a crystalline ionic solid.
2Na + H2 → 2Na + H-
It reacts with H2O to produce H2 gas
2NaH+2H2O → 2NaOH+2H2
Although Na+H- does not conduct electricity in the solid state, the electrolysis of its melt produces H2 at the anode and Na at the cathode.
At cathode At anode
Na + H- (l) &r
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Conductance of a solution depends upon the number of ions present in the solution since sodium chloride is an ionic compound when mixed in water it ionizes completely forming an aqueous electrolytic solution. Hence, the solution conducts electricity while sugar being a non-electrolyte does not ionize in water and when concentration of NaCl is increased, more Na+ and Cl- ions will be produced. Hence, conductance increases.
New Question
10 months agoContributor-Level 10
Kumaun University admission dates for the BA course are out. Moreover, candidates are required to apply for the course soon, as the deadlines are also announced. The BA course is offered via its different affiliated colleges; candidates who complete the basic eligibility for the course can apply for the course and wait for the release of the merit list.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Conjugate acid and conjugate base can be shown as-
HCl(aq) + H2O (l) ? H3O+(aq) + Cl-(aq)
acid base conjugate conjugate
acid base
New Question
10 months agoContributor-Level 10
As of now, the KSOU has not released any application deadline for the MBA programmes. However, interested candidates can visit the official website to apply for their course specialisation. Those who are eligible must provide the relevant documents to complete verification. The applications are available on the KSOU's official admission portal.
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
It is lithium hydride (LiH) because it has significant covalent character due to the smallest alkali metal, LiH is very stable. It is almost nonreactive towards oxygen and chlorine. It reacts with Al2Cl2 to form lithium aluminium hydride.
8LiH+ Al2Cl6 → 2LiAlH4 → LiCl
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: (a) As we know,
ΔG= ?G? +RTlnQ
?G? = Change in free energy as the reaction proceeds,
ΔG = Standard free energy change,
Q = Reaction quotient,
R = Gas constant,
T = Absolute temperature.
Since, ?G? =−RTlnK
∴ΔG=−RTlnK + RTlnQ = RTlnKQ?
If Q
If Q=K, ΔG=0, reaction is in equilibrium and no net reaction is there.
(b) When we increase the pressure equilibrium will shift in forward direction it means Q
Types of Chemical Reactions :
There are 4 main types of chemical
New Question
10 months agoContributor-Level 10
Rajagiri College of Social Sciences provides its students with an option to pursue a BSc (Hons) or a BSc (Hons with Research) in Computer Science with Data Analytics. The duration of these courses is four years, which is further divided into eight semesters. The curriculum for both these courses is the same for seven semesters. The course-wise curriculum during the final (eighth) semester is as follows:
| BSc (Hons) | BSc (Hons with Research) |
|---|---|
|
|
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
Since, a colourless liquid 'A' contains only hydrogen and oxygen and decomposes slowly on exposure to light but is stabilised by addition of urea, therefore, liquid A may be hydrogen peroxide. A is H2O2.
Structure of H2O2 is given below.
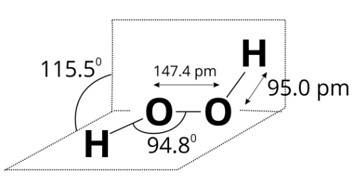
New Question
10 months agoContributor-Level 10
Students should look beyond just fees and salary. Key factors include the specialisation offered, curriculum relevance, industry exposure, internship opportunities, faculty expertise, and placement support. The MBA at DIT University stands out for its unique focus on political leadership and government, real-world learning experiences, and strong placement in political and public policy sectors.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: The equation can be written as-
Ap+x + Bq- y ? xAp+ (aq) + yBq-
S moles of A and B dissolves to give x S moles of Ap+ and y S moles of Bq-.
Ksp = [Ap+]x [Bq-]y = [x5]x [y5]y
= xx y y 5 x+y
New Question
10 months agoContributor-Level 6
To get admission to NIT Jamshedpur, candidate must have a rank as per the cutoff list according to the category they belong to. The cutoff was released for MSc in Physics, MSc in Mathematics and MSc in Chemistry. Among the General AI category, the round 1 cutoff was between 1088 and 1442. Therefore, to get admission into the college through CCMN irrespective of the branch, candidate must have a rank of 1442 or less. The branch-wise cutoff has been given in the table below:
| Course | 2025 |
|---|---|
| M.Sc. in Physics | 1442 |
| M.Sc. in Mathematics | 1088 |
| M.Sc. in Chemistry | 1249 |
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
Mass of H2O2 = 68 g.
Mol. Mass of H2O2=34gmol-1
∴1L of M solution of H2O2 will contain H2O2=34×5 g
∴2L of 5 M solution will contain H2O2=34×5×2=340g
or 200 mL of 5 solution will contain H2O2 = 200 = 34 g
Now 2H2O2 → 2H2O+O2
64 g 32g
Now 68 g of H2O2 on decomposition will give O2=32g
∴34g of H2O2 on decomposition will give O2 = 34 =16 g
New Question
10 months agoContributor-Level 10
Khatima Institute of Technology and Management (KITM Group of Institutions) application process is conducted online. Students can apply online at the official website of the institute or via the Samarth portal. Before proceeding with the application process, students need are advised to check the admission requirements of the desired course. The steps to apply online for Khatima Institute of Technology and Management (KITM Group of Institutions) are as follows:
Step 1: Visit the official website of the college and click on the “Course” tab on the homepage.
Step 2: Alternatively, you can select the "Admission form" option fro
New Question
10 months agoNew Question
10 months ago
Contributor-Level 10
GRE exam is not for students who belong to any specific stream. Graduate Record Examinations is a popular standardized test. It is required for admission to graduate programs. Whether you are planning to pursue PG courses in psychology, economics, or other streams. Many universities accept GRE scores.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: According to Le Chatelier's principle, when we raise the temperature, it shifts the equilibrium to left and decreases the equilibrium concentration of ammonia since it is an exothermic reaction. In other words, low temperature and high pressure is favourable for high yield of ammonia. There will be no change in equilibria on addition of argon (Ar).
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: The values of Kc and Qc are itself sufficient to explain the direction of reaction and less than or greater than one another decides the direction in which reaction will proceed as follows-
(i) As Qc < Kc, the reaction proceeds in the forward direction.
(ii) If Qc > Kc, the reaction will proceed in the direction of reactants (reverse reaction).
(iii) If Qc = Kc, no net reaction occurs.
New Question
10 months agoContributor-Level 10
(i) Industrial preparation: H2O2 is prepared by the auto-oxidation of 2- alkylanthraquinols
2-ethylanthraquinol ↔ H2O2+ oxidised product
2Fe2+ (aq)+2H+ (aq)+H2O2 → Fe3 (aq)+2H2O (l)
PbS (s)+4H2O2 (l) → PbSO4 (s)+4H2O (l)
(ii) Reducing action of hydrogen peroxide
2MnO4-+6H++5H2O2→ 2Mn2++8H2O+5H2
HOCl+H2O2 → H3O++Cl-+O2
Oxidising action of hydrogen peroxide
2Fe2++H2O2→ 2Fe3++2OH-
Mn2++H2O2 → Mn4++2OH-
Acidic properties of H2O2
I2+H2O2+2OH-→ 2I-+2H2O+H2
2MnO4-+3H2O2→ 2MnO2+3O2+2H2O+2OH-
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
