Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
Kb= 5.4×10−4
c= 0.02M
Then, α= (Kb /c)1/2
α= (5.4×10−4 / 2 x 10-2)1/2 =0.1643
(CH3)2NH+H2O ↔ (CH3)2NH+2+OH-
[ (CH3)2NH] = 0.02 – x ≈ 0.02
[ (CH3)2NH+2] = x
[OH-] = 0.1 + x
≈ 0.1
Now, Kb= [ (CH3)2NH+2] [OH−]/ [ (CH3)2NH] = (x × 0.1) / (0.025).
x = 1.08 x 10-4
% of dimethylamine ionised = (1.08 x 10-4) x (100 / 0.02) = 0.54%
New Question
11 months agoContributor-Level 10
Guwahati University accepts a variety of national entrance exams such as JEE Main, GATE, CAT, MAT, XAT, CMAT, and others. Additionally, students with scores of 75 percentile or higher in these national exams may be exempted from the university's written test and proceed directly to the Group Discussion and Personal Interview rounds.
New Question
11 months agoContributor-Level 10
Students willing to enroll into M.Tech courses at SRM University can appear for the SRMJEE PG exam. As per the official website of the university, the SRMJEE PG phase 2 dates are out. As per the schedule, the SRMJEEE PG exam phase 2 was conducted tentatively from Jun 12 to Jun 17, 2025. Registered students must appear on the scheduled exam date.
New Question
11 months agoContributor-Level 10
The total tuition fee of BSc at MMU Sadopur is INR 1.6 Lakh. The BSc programme at MMU Sadopur is four years long and candidates have to pay the tuition fee over that period. Therefore, candidates have to pay an annual fees of INR 40,000 as part of the BSc tuition fees at MMU Sadopur.
New Question
11 months agoContributor-Level 10
pKa? =? logKa= 4.74
Ka? = 10? pKa =10?4.74 = 1.8*10?5
Let x be the degree of dissociation. The concentration of acetic acid solution, C = 0.05 M
The degree of dissociation,
x= (Ka / C)1/2? = (1.8*10?5 / 0.05)1/2 ? = 0.019
(a) The solution is also 0.01 M in HCl.
Let x M be the hydrogen ion concentration from ionization of acetic acid. The hydrogen ion concentration from ionization of HCl is 0.01 M. The total hydrogen ion concentration
[H+] = 0.01 + x
The acetate ion concentration is equal to the hydrogen ion concentration from ionization of acetic acid. This is also equal to the concentration of acetic acid that has dissociated.
[CH3?
New Question
11 months agoContributor-Level 10
The highest stipend offered during IIM Rohtak MBA summer placements for the batch of 2024-26 is presented below:
Particulars | PGP Summer Placement Statistics (2024-26 Batch) |
|---|---|
the highest stipend | INR 3.6 lakh |
New Question
11 months agoContributor-Level 9
Cluster University Srinagar BCA graduates can find various career opportunities in the government and private sectors. The degree holders can work as a web developer, software developer, data analyst, cybersecurity specialist, or network administrator. Often, the UG degree holders opt for higher studies and pursue MCA or MBA to boost their career prospects.
Some other popular job roles for BCA degree holders are as follows:
- Mobile app development
- Cloud computing
- IT consulting
New Question
11 months agoContributor-Level 6
Simple Present Tense is the kind of verb tense which is used in our everyday life. The scenarios where Simple Present Tense can be used are:
- To describe daily routine
- To describe facts or universal truths
- To describe scheduled events
- To describe feelings and state of being
- To describe directions and instructions
- To describe habits
New Question
11 months agoContributor-Level 10
SRM University has commenced the M.Tech admission process for the academic year 2025. Students who wish to enroll into the courses can visit the university's official website and apply. However, the university has not announced any deadline to fill out the application form. Students are advised to register timely to avoid missing out on the application process.
New Question
11 months agoContributor-Level 10
Let c be the initial concentration of C6H5NH3+ and x be the degree of ionisation.
C6H5NH2 + H2O? C6H5NH3+ + OH-
c (1-x) cx cx
Kb = [C6H5NH3+] [ OH-] / [C6H5NH2]
= [cx] [cx] / [c (1 – x)]
Since x is very small and negligible 1 – x≈ 1
∴Kb= [cx] [cx] / [c] = cx2
=> x =
=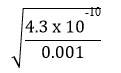
= 6.56 x 10-4
∴ [OH-] = cx = 0.001 x 6.56 x 10-4 = 6.56 x 10-7 M
[H+]= Kw / [OH-] = 10-14 / 6.56 x 10-7 = 1.52 x 10-8
pH= &ndash
New Question
11 months ago
Contributor-Level 10
IPFC Academy offers certification courses such as CMA USA, CIA USA, CPA USA and ACCA UK.The college prepares students through its Online Live Classes. Students with work experience can enroll to these programmes.
New Question
11 months agoContributor-Level 10
pH = 9.95,
pOH = 14 – pH = 14 − 9.95 = 4.05
[OH−] = 10−pOH = 10−4.05 = 8.913 × 10−5
Codeine + H2? O? CodeineH+ + OH−
The ionization constant, Kb? = [CodeineH+] [OH−] / [codeine]?
= [ (8.913×10−5)× (8.913×10−5)] / 5×10−3
= 1.588×10−6.
pKb? = −log (1.588×10−6)
= 5.8
New Question
11 months agoContributor-Level 10
Once the M.Tech selection process is completed at SRMU AP, students with admission offers are required to pay a part of the tuition fees to secure their seat. As per official sources, the total tuition fees for M.Tech courses amount to INR 3 lakh.
Note: The above-mentioned fee is as per the official sources. However, it is indicative and subject to change.
New Question
11 months agoContributor-Level 10
The domain-wise average package received during IIM Rohtak placements 2025 is presented below:
Domain | PGP Average Package (2025) |
|---|---|
Sales & Marketing | INR 17.73 LPA |
Product Management, IT and Operations | INR 19.62 LPA |
Consulting | INR 16.05 LPA |
Finance | INR 17.01 LPA |
General Management and HR | INR 16.16 LPA |
New Question
11 months agoContributor-Level 10
H+] = cα = 0.1 × 0.132 = 0.0132M
pH = −log [H+] = −log0.0132
= 1.88
The acid dissociation constant is
Ka? = cα2? / (1−α) = 0.1 × (0.132)2 / (1−0.132)
= 2.01×10−3.
pKa? = −logKa? = −log (2.01×10−3) ≈ 2.7
New Question
11 months agoContributor-Level 10
(a) For 2g of TlOH dissolved in water to give 2 L of solution:
[TlOH] = [OH−] = (2×1)? / (2×221) = (1 / 221)? M
pOH = −log [OH]− = −log (1/221)?
= 2.35
pH = 14 – pOH = 14 − 2.35 = 11.65
(b) For 0.3 g of Ca (OH)2? dissolved in water to give 500 mL of solution:
[OH−] = 2 [Ca (OH)2? ] = 2 (0.3×1000/500? ) = 1.2M
pOH = −log [OH−] = −log1.2 = 1.79
pH= 14−pOH=14−1.79
=12.21
(c) For 0.3 g of NaOH dissolved in water to give 200 mL of solution:
[OH−]= [NaOH] = 0.3×1000/200? = 1.5M
pOH= −log [OH−] = &mi
New Question
11 months agoContributor-Level 10
The highest package offered during IIM Rohtak placements over the past three years is presented below:
Particulars | PGP Placement Statistics (2023) | PGP Placement Statistics (2024) | PGP Placement Statistics (2025) |
|---|---|---|---|
the highest package | INR 36 LPA | INR 48.25 LPA | INR 36 LPA |
New Question
11 months agoContributor-Level 10
(a) 0.003 M HCl
[H3? O+] = [HCl] = 0.003M
pH = −log [H+] = −log (3.0×10−3) = 2.523
(b) 0.005 M NaOH
[OH−] = [NaOH] = 0.005M
[H+] = Kw? / [OH−]? = 10−14/ 0.005? =2×10−12
pH= −log [H+]=−log (2×10−12)=11.699
(c) 0.002M HBr
[H+]= [HBr]=0.002
pH= −log [H+]=−log0.002=2.699
(d) 0.002M KOH
[OH−]= [KOH]=0.002M
[H+]= Kw / [OH−]? =10−14 / 0.002? =5×10−12
pH= −log [H+]=−log (5×10−12)=11.301
New Question
11 months agoContributor-Level 10
SRM University offers an array of specialisations under its M.Tech programme. At the time of application, students must indicate the choice of specialisation. The following is a list of all the courses offered by the university for students to choose from:
- MTech in Artificial Intelligence and Machine Learning
- MTech in Data Science
- MTech in Cyber Security
- MTech in Embedded Systems and IOT
- MTech in VLSI
- MTech in Materials and Manufacturing Technology
- MTech in Thermal Engineering
- MTech in Environmental and Sustainable Engineering
New Question
11 months agoContributor-Level 6
There are three kinds of simple present tense in the English language. The structure for each is as follows:
Affirmative: Subject + base verb (add -s/-es for third-person singular)
Negative: Subject + do/does + not + base verb
Interrogative: Do/Does + subject + base verb +?
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
