Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (i) and (iv)
When a chalk stick is dipped in ink, the surface retains the colour of the ink due to adsorption of coloured molecules
while the solvent of the ink goes deeper into the stick due to absorption so the answers are (i) and (iv).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (i) and (ii)
Substances which accelerate the rate of a chemical reaction and themselves remain chemically and quantitatively unchanged after the reaction, are known as catalysts they can undergo physical change so the answers are (i) and (ii).
New Question
10 months agoContributor-Level 10
Admission to MDS course is based on entrance at Anil Neerukonda Institute of Dental Sciences. The MDS full form is Master of Dental Surgery. MDS courses are PG degree programs that offer advanced training in dental specialties. The MDS course duration is 3 years. MDS courses open up a range of career opportunities in private practice, academics, and specialised clinical settings, allowing professionals to advance their skills and expertise in the Dental field.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (ii) and (iii).
When electrophoresis, i.e, movement of particles is prevented by some suitable means, it is observed that the dispersion medium begins to move in an electric field. This phenomenon is termed electroosmosis so the answers are (ii) and (iii).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (i) and (iii)
Lyophilic colloids have a unique property of protecting lyophobic colloids. When a lyophilic sol is added to the lyophobicm sol, the lyophilic particles form a layer around lyophobic particles and thus protect the latter from electrolytes. Lyophilic colloids used for this purpose are called protective colloid so the answers are (i) and (iii).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (iii) and (iv)
Sols directly formed by mixing substances like gum, gelatin, starch, rubber, etc., with a suitable liquid (the dispersion
medium) are called lyophilic sols. They are also known as reversible colloid. These sols are very stable and cannot
coagulate easily hence the answers are (iii) and (iv).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (i) and (iv)
Negatively charged emulsion can be precipitated by oppositely charged electrolyte. Na+ and K+ from the electrolyte can neutralize the negatively charge emulsion and precipitate the colloid so the answers are (i) and (iv).
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (ii) and (iv)
Emulsions can be broken into constituent liquids by heating, freezing and centrifuging hence the answer is (ii) and (iv).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (ii) and (iv)
The presence of equal and similar charges on colloidal particles is largely responsible in providing stability to the colloidal
solution, because the repulsive forces between charged particles having same charge prevent them from coalescing or
aggregating when they come closer to one another. The Brownian movement has stirring effect which does not permit the particles to settle and thus, is responsible for the stability of sols hence the answer is (ii) and (iv).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (ii) and (iii)
H2 molecule on an activated charcoal is adsorbed to a very little extent in comparison to easily liquefiable gases because it has (a) Very weak van there Waals force of attraction (b) Very low critical temperature hence the answer is (ii) and (iii).
New Question
10 months agoNew Question
10 months agoNew Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (i) and (iii)
Freundlich gave an empirical relationship between the quantity of gas adsorbed by unit mass of solid adsorbed and pressure at a particular temperature.
=kp
If = 0 ; = k xtent of adsorption is independent of pressure
When n=0 ; = kp
vs p is a line
parallel to x-axis
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (i) and (ii)
The action of a catalyst is selective in nature and so a substance which acts as a catalyst in one reaction may fail to catalyse another reaction. They also do not change the enthalpy of reaction hence the answer is (i) and (ii).
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: (ii) and (iii)
The formation of micelles takes place only above a particular temperature which is the Kraft temperature (Tk ) and above a particular concentration i.e., the critical micelle concentration (CMC). Upon dilution, these colloids revert back to the individual ions hence the answer is (ii) and (iii).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (ii)
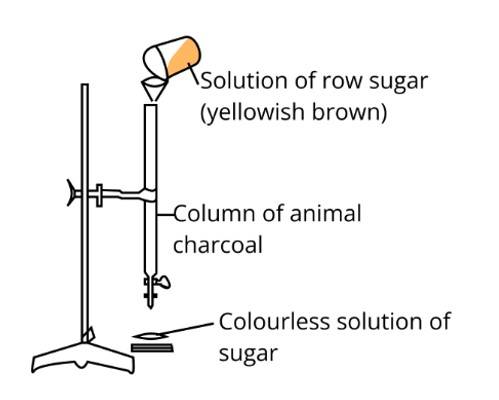
In the figure adsorption of coloured particle from charcoal is shown. Solution of raw sugar is filtered by animal charcoal
and yellowish brown colour of raw sugar is adsorbed and filterate is colourless which gives white colour on crystallization
hence the answer is (ii).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct answer is (iv)
Charge on the sol particles can be a result of the following:
Due to electron capture by sol particles during electrodispersion of metals,
Due to preferential adsorption of ions from solution and/or
Due to formulation of electrical double layer.
Hence the correct answer is (iv)
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts