Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is iii.
River water is a colloidal solution of clay. Sea water contains a number of electrolytes. When river water meets the sea water, the electrolytes present in sea water coagulate the colloidal solution of clay resulting in its deposition with the formation of delta hence the answeris (iii).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct answer is (ii)
The correct sequence of steps involved in catalysis are:
(i) Adsorption of A and B on surface
(ii) Interaction between A and B to form intermediate
(iii) Starting of desorption from surface
(iv) Complete desorption from the surface
Therefore, the correct answer is (ii)
New Question
10 months agoContributor-Level 10
The eligibility for the UG courses at Future Institute of Management Studies is that candidates must have cleared Class 12 with a minimum of 45 to 50% aggregate based on their desired course.
Candidates are required to follow the admission process if they meet the minimum eligibility for the course. Candidates who are eligible must apply for the courses through the application form.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (iv).
Colloidal particles are bigger aggregate than the number of particles in a colloidal solution hence, the values of
colligative are of small order as compared to values shown by true solutions at same concentration so the answer is (iv).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (iv).
Solid and liquid together forms up sol. In this case, solid is dispersed phase and liquid is the dispersion medium hence the answer is (iv).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (ii)
The rate of coagulation will be faster if the value of oppositely charge electrolyte is high hence the answer is (ii).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (iv)
Peptisation is the process in which freshly prepared precipitate can be converted into colloidal solution so the answer is (iv).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (iii).
Lyophilic colloids have unique property of protecting lyophobic colloids. When a lyophilic sol is added to lyophobic solution, the lyophilic particles form a layer around the lyophobic particles and protect the latter from electrolytes. Lyophilic colloids used for this purpose are called protective colloids hence the answer is (iii).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (ii).
Tyndall effect is defined as the optical property shown by colloidal particle. Above the critical micelle concentration, a solution of soap behaves as an associated colloid so it shows Tyndall effect hence the answer is (ii).
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (ii)
There are few substances which at low concentrations behave as normal strong electrolytes, but at higher concentrations exhibit colloidal behavior due to the formation of aggregates. The aggregated particles formed are called micelles. These are also known as associated colloids hence the answer is (ii).
New Question
10 months agoContributor-Level 10
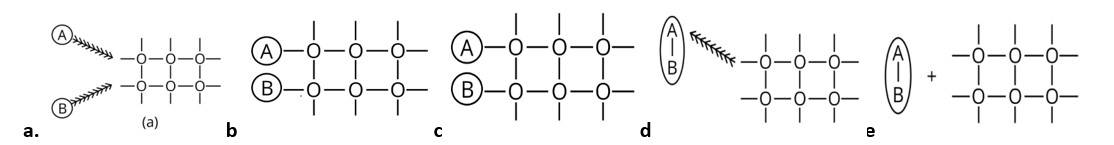
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (i)
When the reactant and catalyst are in different phase it is known as heterogeneous catalysis hence the answer is (i).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (iv)
Lesser the value of critical temperature of gases, the lesser will be the force of attraction among molecules and least will be the adsorption hence the answer is option (iv).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (iii)
In absorption, a substance is uniformly distributed, through the body of the solid or liquid, hence the correct option is (iii).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (i)
Physisorption adsorbent does not show specificity for any particular gas because it is involved van there Waals forces are universal, hence the correct option is (i).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (ii)
On increasing the temperature activation energy of the adsorbate molecule increases which convert physical adsorption into chemisorptions hence the correct option is (ii).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (i)
High temperature is not favourable for physical adsorption since it is an exothermic process, hence the correct option is (i).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (i)
As adsorption is an exothermic process so? H cannot be greater than zero, hence the correct option is (i).
New Question
10 months agoContributor-Level 10
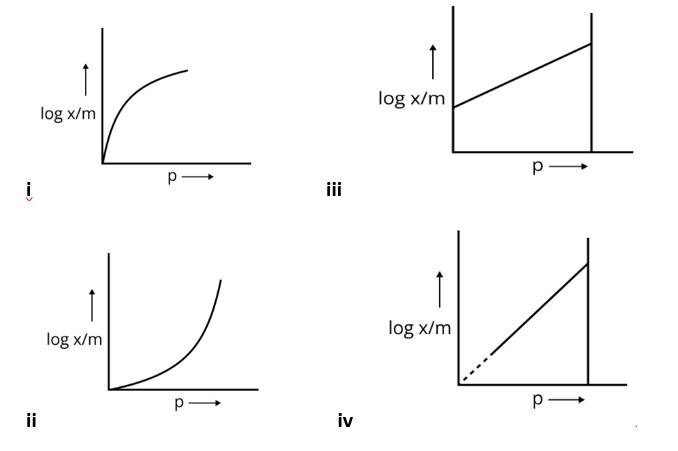
This is a Multiple Choice Type Questions as classified in NCERT Exemplar
Ans: Correct option is (i)
Extent of adsorption depends on the concentration of the solute in a solution. The concentration of adsorbate increases the interaction between adsorbate and adsorbent thus the extent of adsorption also increases; hence the correct option is (i).
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts