Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
(1) Known - E0Cr3+/Cr = - 0.74 V
E0 cd2+ = - 0.40 V
? rG0 =? K =?
The galvanic cell of the given reaction is written as - Cr (s)|Cr3+ (aq)| Cd2+ (aq)|Cd (s)→ Reaction 1
Hence, the standard cell potential is given as, E0 = ER0 - EL0
= - 0.40 - (- 0.74)
∴ E0 = + 0.34 V
To calculate the standard Gibb’s free energy? rG0, we use,
? rG0 = - nE0F → Equation 1
wherenF is the amount of charge passed and E0 is the standard reduction electrode potential. Substituting n = 6 (no. of e - involved in the reaction 1), F = 96487 C mol-1,
E0 = + 0.34 V in Equation 1, we get, l
? rG0 = - 6×0.34V×96487 C mol-1
= - 196833.48 CV m
New Question
11 months agoGuide-Level 15
The total tuition fee for Karnataka State Dr. Gangubai Hangal Music and Performing Arts University BPA is INR 9,000. This information is sourced from official website/ sanctioning body and is subject to change. Candidates need to pay some part of the fee at the time of admission to confirm their seat. The total fee of BPA includes various components such as caution deposit, exam fee, tuition fee, library fee, admission fee, etc.
New Question
11 months agoContributor-Level 10
14.4
Amino acids are organic compounds containing amine (basic) and carboxyl (acidic) functional group with a specific side chain. Both acidic and basic group are present in the same molecule. In, aqueous solution carboxyl group can lose a proton (H+) and amino group can accept a proton (H+) giving rise to the dipolar ion called as zwitter ion. Zwitter ion is shown below:
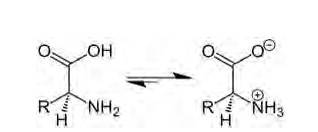
In this zwitter ion there is the presence of both positive as well as negative charge, so there is the development of strong electrostatic force of attraction between the molecules and the water. For this reason solubility of amino acids is higher. Due to strong e
New Question
11 months agoContributor-Level 10
Yes, applications are open at Dfly International Aviation Institute for various courses. The mode of application is online. Aspirants need to apply at the official website to registration to the various programme. Candidates can fill out the application form on the institute's official website.
New Question
11 months agoContributor-Level 10
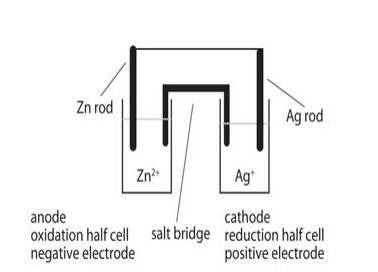
The galvanic cell corresponding to the given redox reaction can be represented as:
Zn|Zn2+ (aq)|Ag + (aq)|Ag
- 1) Zn electrode (anode) is negatively charged because, at this electrode, Zn is oxidized to Zn2+, causing electron accumulation at the
- 2) Electrons (ions) are the carriers of the current in the cell and in the external circuit, current flows from Ag (cathode) to Zn (anode) which is normally opposite to the electron flow which is from anode to cathode.
- 3) At anode:
Zn (s)⇒ Zn2 + (aq) + 2e– At cathode:
Ag + (aq) + e –⇒ Ag (s)
New Question
11 months agoContributor-Level 10
Some of the most popular BCA colleges in Karnataka where students can study include colleges like- Christ University, Kristu Jayanti College, Jain Deemed-to-be University, Mount Carmel College, Presidency College, St. Aloysius College, etc.
New Question
11 months agoContributor-Level 10
K + /K = –2.93V, Ag+ /Ag = 0.80V, Hg2+/Hg = 0.79V Mg2+/Mg = –2.37 V, Cr3+/Cr = – 0.74V
A 3.2 Reducing power of metals increase with the decrease of reduction potential. Hence, the increasing order of reducing power will be as,
Ag < Hg < Cr < Mg < K
When the reduction potential is lower, the element has more tendency to get oxidized and thus more will be reducing power. The metal that has more negative electrode potential will be the one with more reducing power. Thus, here potassium (K) has the highest reducing power among the given elements.
New Question
11 months agoContributor-Level 10
14.3
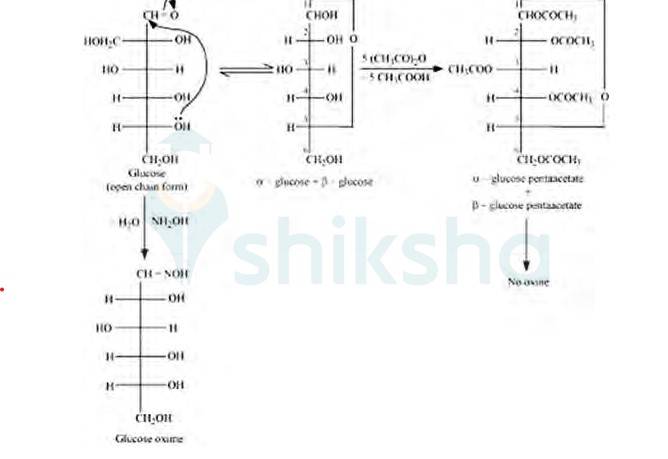
D-glucose reacts with hydroxylamine (NH2OH) to form oxime due to the presence of the aldehyde functional group (-CHO). This is due to the cyclic structure of glucose which forms an open chain structure in an aqueous medium, which then reacts to give an oxime.
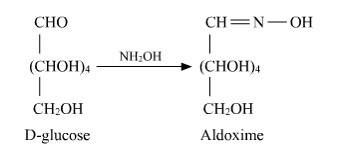
But in case of pentaacetate of D-glucose, it does not form open chain structure in an aqueous medium so it does not react with NH2OH.
New Question
11 months agoContributor-Level 10
In order to get admission at Dfly International Aviation Institute candidates must keep certain documents ready. Student can check the list of documents below:
- Scanned Class 10 and Class 12 marksheet
- Leaving Certificate
- Aadhaar Card
- Passport-size photograph
- Signature
Note: The list of documents mentioned above are the general documents. For more details visit the official website.
New Question
11 months agoContributor-Level 10
The order in which the given metals displace each other from the solution of their salts is given by,
Mg>Al> Zn> Fe> Cu
A metal of stronger reducing power displaces another metal of weaker reducing power from its solution of salt. The order of increasing the reducing power of given metals is Cu< Fe< Zn< Algiven metals displace each other from the solution of their salts is given by, Mg>Al> Zn> Fe> Cu. This is hence arranged in decreasing order of its reactivity
New Question
11 months agoBeginner-Level 4
In order to be admitted into the MCA programme at PICT, Pune, you must first appear and pass the MAH MCA CET or PERA CET, as well as the NIMCET test. In addition, you must have a Bachelor's degree with at least 50% grades (45% for reserved categories). Specific eligibility requirements and entrance tests may change from year to year, thus the most up-to-date information can be found on the official PICT website.
New Question
11 months agoContributor-Level 6
SITASRM Institute of Management and Technology is a private institute located in Greater Noida, Uttar Pradesh. The institute is recognised by the All India Council for Technical Education (AICTE) and is affiliated with AKTU. SIMT is a well-reputed institute that offers courses across the Management and Computer Applications streams at the UG and PG levels. The courses include BCA, BBA, MCA and MBA in full-time mode.
New Question
11 months agoContributor-Level 10
There are around 25 colleges in Karnataka that offer the BCA course. This includes 24 private BCA colleges in Karnataka. Students can take admission to these colleges based on their entrance exam scores in the Christ University Entrance Exam (CUET), JET, KCET, etc.
New Question
11 months agoContributor-Level 10
In the corrosion reaction, due to the presence of air and moisture, oxidation takes place at a particular point of an object made of iron. That spot behaves as the anode. The reaction at the anode is given by,
Fe (s) ⇒ Fe2+ (aq) + 2e-
Electrons released at the anodic spot move through the metal and go to another spot of the object, wherein presence of H+ ions, the electrons reduce oxygen. This spot behaves as the cathode. These H+ ions come either from H2CO3, which are formed due to the dissolution of carbon dioxide from the air into water. The cathodic reaction is given by
O2 (air) + 4Haq++4e-⇒ 2H O
The overall reaction is give
New Question
11 months agoContributor-Level 10
Suggest two materials other than hydrogen that can be used as fuels in fuel cells.
New Question
11 months agoGuide-Level 15
Candidates who have passed PUC/ equivalent/ JOC/ two-year Diploma can apply for BPA course at Karnataka State Dr. Gangubai Hangal Music and Performing Arts University. Aspirants must hold the documents proving their eligibility for verification at the time of admission. Besides, candidates must also appear for the common entrance test conducted by the university for BPA admission.
New Question
11 months agoContributor-Level 10
Anode: Lead (Pb)
Cathode: a grid of lead packed with lead oxide (PbO2)
Electrolyte: 38% solution of sulphuric acid (H2SO4)
The cell reactions are as follows :
Pb (s) + SO2-4 (aq) ⇒ PbSO4 (s) + 2e- (anode)
PbO2 (s) + SO2-4 (aq) + 4H+ (aq) +2e-⇒ PbSO4 (s) +2H2O (l) (cathode)
Pb (s) + PbO2 (s) +2H2SO4 (aq)⇒ 2PbSO4 (s) +2H2O (l)
(overall cell reaction)
On charging, all these reactions will be reversed.
New Question
11 months agoContributor-Level 10
Cr2O72– + 14H+ + 6e–⇒ 2Cr3+ + 7H2O
A 3.12
Cr2O72– + 14H+ + 6e–⇒ 2Cr3+ + 7H2O
For reducing one mole of Cr2O72–, 6 mole of electrons are required. Hence, 6 Faraday charges is needed. Hence, 6F = 6×96487 = 578922 C. Thus, the quantity of electricity is needed is 578922 C.
New Question
11 months agoContributor-Level 10
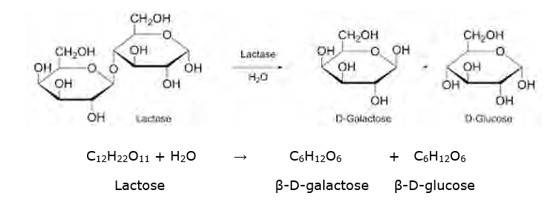
14.2
Lactose is a disaccharide carbohydrate (made up of two monosaccharide units) composed of β-D-galactose and β-D-glucose units. Hydrolysis breaks the glycosidic bond converting sucrose into β-D- galactose and β-D-glucose.
NOTE: But however, this reaction is so slow that it takes years for the solution of sucrose to undergo negligible change. Hence an enzyme called sucrase is added to proceed rapidly.
New Question
11 months agoGuide-Level 15
Candidates applying for admission at Karnataka State Dr. Gangubai Hangal Music and Performing Arts University have to pay an application fee. Aspirants can pay the fee via any of the online payment modes. Below is the application fee for a few courses offered by the university:
| Course | Application Fee (General) | Application Fee (SC/ ST/ Cat-I) |
|---|---|---|
| PhD/ D.litt | INR 2,000 | INR 1,000 |
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
