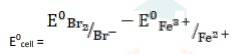Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
14.16
Starch consists of two components – amylase and amylopectin. Amylose is a long linear chain of α–D-(+)-glucose units joined by C1-C4glycosidic linkage (α -link).
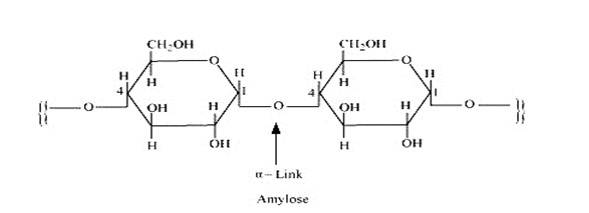
Amylopectin is a branched-chain polymer of –D-glucose units, in which the chain is formed by C1-C4 glycosidic linkage and the branching occurs by C1-C6 glycosidic linkage.

On the other hand, cellulose is the main structural material of tree and other plants. Wood is 50% cellulose, while cotton wool is almost pure cellulose. It is linear chain natural polymers of β-D-glucose units joined by 1, 4-glycosidic linkage (natural linear polymers).
New Question
11 months agoContributor-Level 10
Sri Krishnadevaraya University MBA admission applications for the academic session 2025-27 is open. Candidates who are interested can complete the application form to apply for the MBA programme. Besides, the selection of candidates depends upon their Andhra Pradesh ICET scores. The following are important for entrance exams for MBA admission at SK University:
- AP ICET registration process
- AP ICET exam
- AP ICET Mock Test
- AP ICET Result
New Question
11 months agoContributor-Level 10
The allocation of BDes seats at NIFT Mumbai is subject to rules of reservation set by the institute. As per NIFT's reservation policy, 22.5% of the seats are reserved for the admission of SC/ST category students to its BDes course. Out of which, 15% of seats are reserved for SC students and 7.5% are reserved for the admission of ST students.
New Question
11 months agoContributor-Level 10
The electrode reaction is written as,
2Fe3+ + 2I - → 2Fe2+ + I2
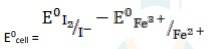
= 0.54V - 0.77V
∴ E0cell = - 0.23 V
It is not feasible, as E0cell is negative, ∴ ?G0 is positive.
- The electrode reaction is written as,
- 2Ag+ (aq) + Cu(s)→ Cu2+ (aq) + Ag(s)
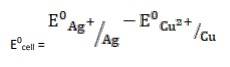
= + 0.80V - 0.34V
∴ E0cell = 0.46V
It is feasible, as Ecell 0 is positive, ∴ ?G0 is negative.
- (iii) The electrode reaction is written as,
- 2Fe3+ (aq) + 2Br- (aq)→ 2Fe2+ (aq) + Br2
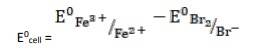 k
k= 0.77V - 1.09V
∴ E0cell = - 0.32 V
It is not feasible, as E0cell is negative, ∴ ?G0 is positive.
- (iv) The electrode reaction is written as,
- Ag(s) + Fe3+ (aq) → Fe2+ (aq) + Ag
New Question
11 months agoContributor-Level 8
Master of Global Business is one of the best courses offered at SP Jain School of Global Business. With the help of this course, students get to develop communication skills, analytical and strategic thinking abilities, and leadership skills by getting to work in teams. Mentioned below are the details of the SP Jain SGM Master of Global Business:
| Particulars | Details |
|---|---|
Annual Tuition Cost | INR 31 L - 34 L |
| Program Duration | 16 months (includes a 4-month internship) |
| Study Location | On-campus in Singapore and Dubai (with an option to study in London at SPJ London) |
| Upcoming Intake | Jun 2025 |
| Specializations/Electives | Global Marketing Management Global Logistics and Supply Chain Management Global Finance Digital Business Management |
the highest Salary | USD 46,254 (INR 39.4 L) |
Average Salary | USD 24,777 (INR 21.1 L) |
Conversion Rate: 1 USD = INR 85.29
New Question
11 months agoContributor-Level 10
Equivalent weight is Ag, EAg = 180/1 = 180
Equivalent weight is Cu, ECu = 63.5 / 2 = 31.75
Equivalent weight is Zn, EZn= 65/2 = 32.5
Using Faraday’s second law of electrolysis, to find the mass of Cu and Zn, we use Equation 1,
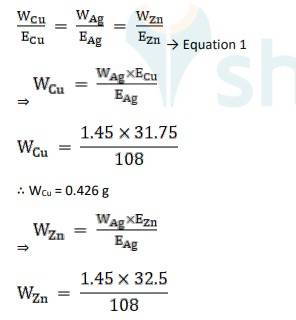
∴ WZn = 0.436 g
To find the time of current flow, using Faraday’s first law of electrolysis we get,
M = Z ×I ×t ⇒ Equation 2
? Z = Equivalent Weight / 96487, Equation 2 becomes,
M = 108 / 96487 X 1.5 X t
t = 1.45 X 96487 / 108X 1.5
t = 864 seconds.
The time of current flow, t = 864 seconds, the mass of Cu is 0.426 g and mass of Zn is 0.436 g
New Question
11 months agoContributor-Level 10
14.15
Hydrolysis is the process of using water to break down a molecule into two parts. It is usually a type of decomposition reaction where one reactant is water, where water is used to break chemical bonds in the other reactant. It can be considered as reverse of a condensation reaction.
The general formula of a hydrolysis reaction is:
XY + H2O → XH + YOH
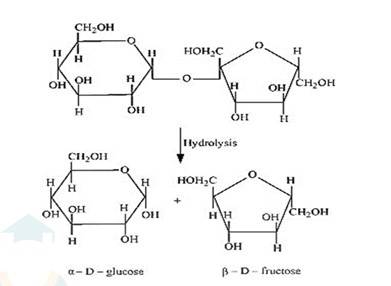
(i) On hydrolysis with dilute acids, sucrose yields an equimolecular mixture of α –D glucose and β–D- fructose.
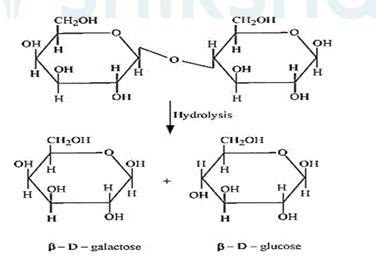
(ii) The hydrolysis of lactose gives β–D-galactose and β–D-glucose as final products.
New Question
11 months agoContributor-Level 10
Quantity of electricity passed = 5 A × (20 × 60 sec)
= 6000 C ⇒ Equation 1
The electrode reaction is written as,
Ni2+ + 2e → Ni
Thus, the quantity of electricity required = 2F
= 2×96487 C
= 192974 C
? 192974 C of electricity deposits 1 mole of Ni, which is 58.7 g ⇒ Equation 2
Thus, equating equations 1 and 2, we get
192974 C of electricity deposits = 58.7 g
6000 C of electricity will deposit = 58.7 X 6000 / 192974
= 1.825g of Ni
The mass of Ni deposited at the cathode is 1.825g of Ni
New Question
11 months agoContributor-Level 10
National Institute of Fashion Technology Mumbai (NIFT Mumbai) offers a total of 329 seats under its BDes programme. This implies that 329 students can be admitted to BDes courses in a single academic year. Moreover, students must note that the stated seat intake is as per official sources. However, it is subject to change.
New Question
11 months agoContributor-Level 10
Unitec Institute of Technology offers around 32 undergraduate programs, 9 Masters programs and 3 post graduate programs to international students to choose from. The UG tuition fee range is INR 3.6 L - 15.09 L and PG tuition fee range is INR 11.05 L - 16.33 L.Tuition fees can differ depending on the specific course selected. Listed below is a table of tuition cost for some popular programs opted by applicants:
Courses | 1st Year Tuition Fees |
|---|---|
MBA/PGDM (2 courses) | INR 14 L - 16 L |
MS (1 course) | INR 16 L |
B.E. / B.Tech (7 courses) | INR 13 L - 15 L |
BBA (3 courses) | INR 11 L |
M.A. (2 courses) | INR 14 L - 16 L |
PG Diploma (6 courses) | INR 11 L - 16 L |
M.Arch (3 courses) | INR 14 L - 16 L |
B.Sc. (5 courses) | INR 12 L - 13 L |
B.Des (1 course) | INR 12 L |
B.A. (1 course) | INR 12 L |
MIM (2 courses) | INR 14 L - 16 L |
B.Arch (2 courses) | INR 13 L |
New Question
11 months agoContributor-Level 10
Yes, candidates can join Dfly International Aviation Institute after Class 12. DIAI Dehradun offers BBA and BCom courses at the UG level. Apart from the degree courses, the college also offers Online programmes, Cabin Crew courses, Travel & Toursim courses, etc. The minimum eligibility for course admissions is to pass Class 12.
New Question
11 months agoContributor-Level 10
14.14
Glycogen is a polysaccharide-type of carbohydrate. In animals, carbohydrates are stored as glycogen.
But starch is a carbohydrate which consists of two components –amylase (15 -20 %) and amylopectin (80 – 85%). However, glycogen is also like amylopectin but branching will take place after every 5 to 6 glucose unit. Also, glycogen is highly branched.
New Question
11 months agoContributor-Level 10
(i) The electrode reaction for 1 mole of H2O is given as,
H2O → H2 + 1/2O2
i.e., O2- →1/2 O2 + 2e -
∴ The quantity of electricity required = 2F
= 2×96487 C
= 192974 C
The quantity of electricity required in coulomb for the oxidation of 1 mol of H2O to O2 is 192974 C
(ii) The electrode reaction for 1 mole of FeO is
FeO + 1/2 O2 → 1/2 Fe2O3
i.e., Fe2+ → Fe3+ + e -
∴ The quantity of electricity required = 1F
= 1×96487 C
= 96487 C
The quantity of electricity required in coulomb for the oxidation of 1 mol of FeO to Fe2O3 is 96487 C
New Question
11 months agoGuide-Level 15
Karnataka State Dr Gangubai Hangal Music and Performing Arts University admission to PhD courses is based on entrance exam score of the candidates. Aspirants with a valid score in JRF/ NET/ KSET can get admission in PhD. Candidates without a score in any of the accepted entrance exams have to appear for the test conducted by the university. Music University conducts an entrance test for 120 minutes. The test is of 100 marks. The syllabus for entrance test shall consist of 10% questions from research
methodology and 90% shall be subject specific.
New Question
11 months agoContributor-Level 10
Students can calculate the ROI offered by various BCA colleges in Karnataka by using the data provided below:
College Name | Average Fees | Average Package |
|---|---|---|
INR 7.73 Lakh | INR 5.16 LPA | |
INR 3.55 Lakh | INR 6.50 LPA | |
INR 6.15 Lakh | INR 5.20 LPA | |
INR 2.87 Lakh | INR 3.33 LPA | |
INR 3.30 Lakh | INR 4.80 LPA |
Disclaimer: This information is sourced from the official website and may vary.
New Question
11 months agoContributor-Level 10
Yes, you can. While CAT, XAT, and other national-level scores are accepted, candidates can also apply through the NU-MAT, the university's own entrance exam. This allows applicants without CAT, XAT, or CMAT scores to still be considered for admission.
New Question
11 months agoContributor-Level 10
There are various BCA colleges in Karnataka that offer great placement opportunities for students. The table below can be used to get an idea about the placement salaries and the top recruiting companies that hire graduates from the BCA colleges in Karnataka.
College Name | Average Placement Salary | Top Recruiters |
|---|---|---|
INR 5.16 LPA | Becton Dickinson, EY GDS, Godrej Properties, etc. | |
INR 6.50 LPA | TCS, Infosys, Amazon, Wipro, Tech Mahindra, IBM, HCL, etc. | |
INR 5.20 LPA | Samsung, Siemens, Schneider, Silicon Labs, TATA Steel, TCS, TATA Technologies, etc. | |
INR 3.33 LPA | DE Shaw, Deloitte, Ernst & Young, Goldman Sachs, HSBC, etc. | |
INR 4.80 LPA | Accenture, Cognizant, HP, HCL, Infosys, etc. |
Disclaimer: This information is sourced from the official website and may vary.
New Question
11 months agoContributor-Level 10
(i) Ca2+ + 2e- → Ca
⇒ Here, 1 mole of Ca, i.e., 40g of Ca requires = 2 F electricity (F if Faraday)
∴ 20g of Ca requires = 20X2/40
= 1 F of electricity
Electricity in terms of Faraday required to produce 20.0 g of Ca from molten CaCl2 is 1 F of electricity.
(ii) Al3+ + 3e- → Al
⇒ 1 mole of Al, i.e., 27g of Al requires = 3 F electricity (F if Faraday)
∴ 40.0 g of Al will require = 3/27 X 40
= 4.44 F of electricity
Electricity in terms of Faraday required to produce 40.0 g of Al from molten Al2O3 is 4.44 F of electricit
New Question
11 months agoContributor-Level 10
The total tuition fee for the two-year MBA programme is approximately INR 30,800. Please note that this amount is based on official sources and may be subject to change. Additional charges may apply as per university guidelines.
New Question
11 months agoContributor-Level 6
To download the Rajasthan Police Constable admit card 2025, candidates need to enter the details shared below-
- SSO ID: The ID that was generated during the registration process.
- Password: The password that was generated by you during the application process.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts