Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoNew Question
11 months agoContributor-Level 8
There are two NDA full forms. One is National Defence Academy which is situated at Khadakwasla, Pune, Maharashtra. This is the military school in India where cadets undergo training before induction in Army, Navy and Air Force. Admission in National Defence Academy is done for Army, Navy and Air Force wings. Through NDA exam candidates are selected for Indian Naval Academy courses. Another NDA full form is National Democratic Alliance.
New Question
11 months agoContributor-Level 10
If there is one or more years of gap between the year of passing class XII exam and applying for admission at AIL, it is considered a Gap year. AFFIDAVIT for Gap Year, should be provided to the exam authorities during the counselling process.
New Question
11 months agoContributor-Level 9
Yes, there are many institutes offering MBA in Advertising course online:
| Online MBA in Advertising Colleges | Tuition Fee |
|---|---|
| Indian School of Communication | INR 5.45 Lakh |
| Xaviers Institute of Business Management Studies | INR 50,900 |
| Indian School of Business Management and Administration | INR 1.92 Lakh |
Note: The information is sourced from the official/counselling body of the institute/exam.
New Question
11 months agoContributor-Level 10
An Air Hostess course trains students to work as cabin crew members in airlines. The Airhostess course teaches grooming, communication skills, safety procedures, and customer service. In the Air Hostess course, students also learn how to handle emergencies and take care of passengers during flights. Airhostess courses are available in the form of Diploma, Certificate, or Degree courses. Students can check the list of reasons to pursue certificate course from Aptech Aviation Academy:
- Professional Training: Learn in-flight safety, customer service, and communication skills.
Grooming and Personality Development: Improve appearance, con
New Question
11 months agoNew Question
11 months agoNew Question
11 months agoContributor-Level 10
Temperature inside the refrigerator, = 9 = 9 + 273 K = 282 K
Room temperature, = 36 = 36 + 273 = 309 K
Coefficient of performance = = = 10.44
Therefore, the coefficient of performance is 10.44
New Question
11 months agoContributor-Level 10
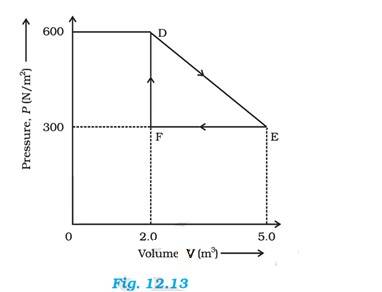
Total work done by the gas from D to E to F = Area of = EF
Where DF = Change in pressure = 600 – 300 = 300 N/
FE = change in volume = 5-2 = 3
Area of 3 = 450 J
Therefore work done by the gas from D to E to F is 450 J.
New Question
11 months agoContributor-Level 10
Heat is supplied to the system at a rate of 100W
Hence, heat supplied, Q = 100 J/s
The system performs at the rate of 75 J/s
Hence, work done, W = 75 J/s
From the 1st law of Thermodynamics, we have Q = U + W, where U is the internal energy
U = Q – W = 100 – 75 = 25 J/s = 25 W
Therefore the internal energy of the given electric heater increases at a rate of 25 W.
New Question
11 months agoNew Question
11 months agoContributor-Level 9
Yes, a few online educational platforms provide free online advertising courses with Certificates:
| Fee Online Platforms | Course Offered |
|---|---|
| Coursera | CU Boulder - Search and Display Advertising |
| Hubspot Academy | Digital Advertising Training Course |
| Great Learning | CU4 to Advertising |
Note: The information is sourced from the official/counselling body of the institute/exam.
New Question
11 months agoContributor-Level 10
Work done by the steam engine per minute, W = 5.4 J
Heat supplied by the boiler, H = 3.6 J
Efficiency of the engine, = = = 0.15
Amount of heat wasted = Input energy – Output energy
= 3.6 5.4 J
New Question
11 months agoNew Question
11 months agoContributor-Level 10
(a) When the stopcock is opened, the volume became double between cylinders A and B. Since volume is inversely proportional to pressure, the pressure will become half. So the initial pressure of 1 atm in cylinder A will become ½ atm in cylinder A and B.
(b) The internal energy will change when there is work done by the gas. In absence of any work done, there will be no change in internal energy.
(c) In absence of any work done, there will be no change in the temperature.
(d) The given process is a case of free expansion. It is rapid and cannot be controlled. The intermediate states do not satisfy the gas equation an
New Question
11 months agoNew Question
11 months agoContributor-Level 10
Aptech Aviation Academy admissions registration form is available at the official website. Interested candidates can fill up the from. The online application process is explained in the following steps:
Step 1: Visit the official Aptech Aviation Academy website (aviationacademydehradun.com).
Step 2: Fill up the form.
Step 3: Complete the form and submit it.
New Question
11 months agoContributor-Level 10
For BA LLB program, following Self Attested Copies of Certificates to be Uploaded along with Online Application Form:
(i) Matric Certificate showing Date of Birth.
(ii) 10+2 Certificate with Detailed Marks Sheet.
(iii) Indian Army/Indian Navy/Indian Air Force Category Certificate as applicable.
(iv) Gallantry Award Certificate (Applicable to Wards of Army Personnel).
(v) Gap Year Affidavit.
New Question
11 months agoContributor-Level 10
The work done, W = 22.3 J
Being an adiabatic process, Q = 0
W = -22.3 J : since the work is done on the system
From the 1st law of thermodynamics, we know Q = W, where is the change of internal energy of the gas
U = 22.3 J
When the gas goes from state A to state B via a process, the net heat absorbed by the system is:
Q = 9.35 cal = 9.35 J = 39.1765 J
Heat absorbed Q = W
W = Q - = 39.1765 – 22.3 = 16.8765 J
Therefore, work done by the system is 16.8765 J
New Question
11 months agoContributor-Level 10
Yes, applications are open at Aptech Aviation Academy for various courses. The mode of application is online. Aspirants need to apply at the official website to registration to the various programme. Candidates can fill out the application form on the institute's official website.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts