Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoBeginner-Level 5
There are various government and private colleges that participate in the Telangana NEET counselling process. The name of the some of the government colleges are given below:
- Gandhi Medical College, Musheerabad, Secunderabad
- Kakatiya Medical College, Warangal
- Rajiv Institute of Medical Sciences, Adilabad
- Government Medical College, Nizamabad
- Government Medical College, Mahabubnagar
Applicants who want to take admission in their desired college are advised to make a list of their preferred choices.
New Question
10 months agoContributor-Level 10
Werner Heisenberg a German physicist in 1927, stated uncertainty principle which is the consequence of dual behaviour of matter and. It states that it is impossible to determine simultaneously, the exact position and exact momentum (or velocity) of an electron.
It rules out existence of definite paths or trajectories of electrons and other similar particles.
New Question
10 months agoContributor-Level 10
The placement rate for National Institute of Management and Technology stood at 92% in 2023. Check out the table below to know more:
Particulars | Placement Statistics (2021) | Placement Statistics (2022) | Placement Statistics (2023) |
|---|---|---|---|
Placement Rate | 74% | 88% | 92% |
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(ii) R–Cl< R–Br < R–I
The reduction of alkyl halides depends upon the C-X bond strength i.e. lesser the bond strength higher will be the reactivity of alkyl halide. As down the group size of the halogens increases thus the bond strength decreases.
New Question
10 months agoContributor-Level 10
The chemical properties of an atom are controlled by the number of electrons in the atom, which remains the same despite the change or variation in atomic masses in case the of isotopes.
New Question
10 months agoContributor-Level 10
The highest package offered during National Institute of Management and Technology placements 2023 stood at INR 18.75 LPA. The table below presents the same:
Particulars | Placement Statistics (2023) |
|---|---|
the highest package | INR 18.75 LPA |
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(i) I2< Br2< Cl2< F2
The reactivity of halogens with alkanes depends on the electronegativity i.e. higher the electronegativity higher would be the reaction. As down the group reactivity decrease so thus its reactivity.
New Question
10 months agoContributor-Level 10
The placements at National Institute of Management and Technology are good. The following table presents the placement statistics for National Institute of Management and Technology.
Particulars | Placement Statistics (2023) |
|---|---|
Placement Rate | 92% |
No. of students placed | 1299 |
the highest package | INR 18.75 LPA |
Average package | INR 5.40 LPA |
PPO's Offered | 452 |
No. of Offers | 1397 |
No. of Companies Visited | 57 |
New Question
10 months agoNew Question
10 months agoContributor-Level 10
For admission to its BArch programme, the Nehru School of Architecture accepts NATA results. The NATA cutoff percentile for candidates in the General All India category was less than 90, following the previous year Nehru School of Architecture cutoffs. A candidate with a 90 percentile can thus be admitted to Nehru School of Architecture based on the cutoff pattern from prior years.
New Question
10 months agoContributor-Level 10
Yes, candidates will have to have their admit card during the exam. Without the admit card, entry to the exam hall will not be allowed. Along with that, a valid ID proof will have to be carried as well to the exam hall.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(iv) C > B > D > A
The boiling point increases with the increase in the length of the side chain. However, with the branching, the boiling point decreases due to the decrease in the surface area.
New Question
10 months agoContributor-Level 10
Sasurie College of Engineering BTech admission forms for 2025 are open for the new academic year, and candidates who want to take admission to any of the BTech specialisations offered at Sasurie College of Engineering can apply online for BTech admission.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The reaction is
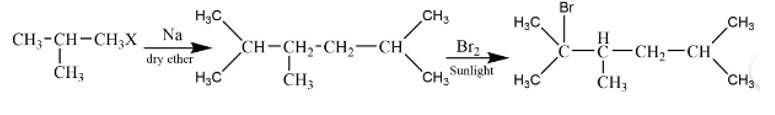
The alkane is
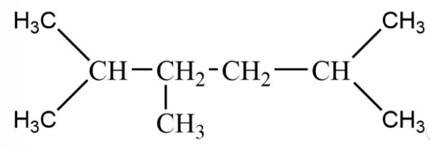
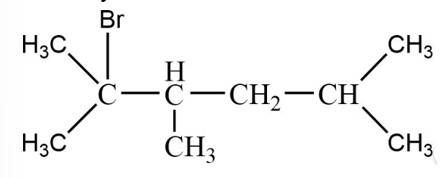
The tertiary bromide is
New Question
10 months agoContributor-Level 10
(a) As Fe3+ contains 5 impaired electrons while Fe2+ contains only 4 unpaired electrons. Fe3+ is more paramagnetic.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
