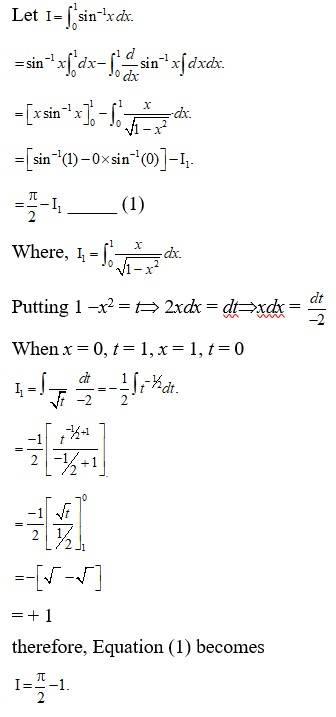Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
Course wise placements for Sasurie College of Arts and Science have not been released yet. However, students can refer to the overall number of students placed and the highest salary given in the table below:
Particulars | Placement Statistics (2024) |
|---|---|
No. of students placed | 182 |
Highest Package | INR 3.50 LPA |
Lowest Package | INR 2 LPA |
Companies visited | 10 |
New Question
10 months agoContributor-Level 10
(a) For n = 4, No. of sub-shells = (l = 0, l = 1, l = 2, l = 3) = 4.
(b) Total number of orbitals which can be present = n2 = 42 = 16.
Each orbital can have an electron with ms = – 1/2. Total no. of electrons with m, = – 1/2 is 16.
New Question
10 months agoContributor-Level 10
The BBA course offered by Sasurie College of Arts and Science BBA runs for a duration of three years. During this course duration, students get to study various subjects related to the field of Management. Some of these subjects are Human Resources Management, Financial Management, Cost Accounting, Basics of Marketing, and more. Those who wish to pursue this course can submit their application on the university's official website.
New Question
10 months agoBeginner-Level 5
Telangana NEET counselling 2025 process comprises various steps. It is being conducted by Kaloji Narayana Rao University of Health Sciences, Telangana. The steps by steps process is given below:
Registration
Choice Filling & Locking
Provisional Allotment
Final Allotment
Merit List
Reporting to the college
New Question
10 months agoContributor-Level 10
Let
Putting ex = t
exdx = dt.
= tan- 1 t + c
= tan- 1 (ex) + c
therefore, Option A is correct.
New Question
10 months agoContributor-Level 10
(a) P (Z=15) : [Ne]103s23p3 No. of unpaired electrons = 3
(b) Si (Z=14) : [Ne]103s23p2 No. of unpaired electrons = 2
(c) Cr (Z=24): [Ar]184s13d5 No. of unpaired electrons = 6
(d) Fe (Z=26): [Ar]184s23d6 No. of unpaired electrons = 4
(e) Kr (Z=36) : [Ar]184s23d104p6 No. of unpaired electrons = Nil.
New Question
10 months agoContributor-Level 10
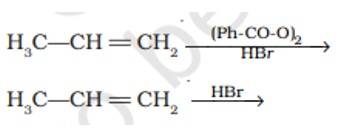
This is a short answer type question as classified in NCERT Exemplar
In presence of (Ph-CO-O)2it lead to CH3-CH2-CH2Br as the reaction undergo by free radical mechanism
However in absence of (Ph-CO-O)2 the reaction undergo by carbocation intermediate thus lead to give CH3-CHBr-CH3
New Question
10 months agoContributor-Level 10
To be able to enroll in the Sasurie College of Arts and Science BBA program, students must follow the below-mentioned steps:
1. Ensure that all the eligibility conditions are met.
2. Students who are eligible can apply by visiting the institute's official website.
3. If allocated a seat, report for the document verification process.
4. Pay the seat acceptance fees.
New Question
10 months agoContributor-Level 10
At Sasurie College of Arts and Science, there are a total of 50 seats available for the BBA course. These seats are allocated based on merit obtained by students in the qualifying examination. Students must note that this seat intake is taken from official sources. However, it is subject to change on a yearly basis at the discretion of concerned authorities.
New Question
10 months agoContributor-Level 10
Configuration of the two elements are:
Al (Z = 13): [Ne]103s23p1
Si (Z = 14): [Ne]103s23p2
The unpaired electrons in silicon (Si) will experience more effective nuclear charge because the atomic number of the element Si is more than that of Al.
New Question
10 months agoContributor-Level 10
Various companies visit Sasurie College of Arts and Science for placements some of them are given below:
Sasurie College of Arts and Science Placement: Top Recruiters | |
|---|---|
KGISL | Lotus |
TCS | Aram Logistics Solutions PVT LTD |
Brand Venture | I Net Resource |
Literact | Face Prep |
New Question
10 months agoNew Question
10 months agoContributor-Level 10
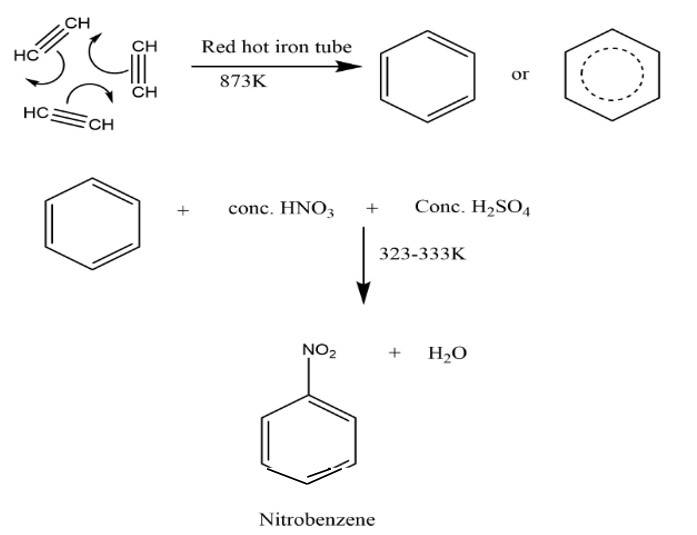
This is a short answer type question as classified in NCERT Exemplar
New Question
10 months agoContributor-Level 10
The total fee for BTech courses at Sasurie College of Engineering includes multiple components, such as tuition fees, admission fees, and various other charges. The total tuition fee for a B.Tech. course ranges from INR 1.6 Lacs to INR 2 lakh. The fee amount mentioned is sourced from the official website/sanctioning body and is subject to change. Therefore, it is indicative.
New Question
10 months agoContributor-Level 10
The average package for Sasurie College of Arts and Science is not out yet. However students can refer to the highest and average package provided in the table below:
Particulars | Placement Statistics (2024) |
|---|---|
the highest Package | INR 3.50 LPA |
the lowest Package | INR 2 LPA |
New Question
10 months agoContributor-Level 10
Greater the penetration of the electron present in a particular orbital towards the nucleus more will be the magnitude of the effective nuclear charge. Based upon this
(i) 2s orbital is closer to the nucleus than 3s orbital and hence it will experience more effective nuclear charge.
(ii) 4d orbital will experience more effective nuclear charge due to its closer proximity to 4f orbital.
(iii) 3p orbital will experience more effective nuclear charge as it is closer to the nucleus
New Question
10 months agoRegister to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts