Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (iv)
MnO2 ( Black )+4HCl→MnCl2+2H2O+Cl2
Greenish yellow color
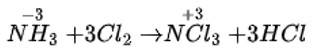
New Question
10 months agoUsing the definition, prove that the function is invertible if and only if is both one-one and onto.
Contributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (iii)
C+2H2SO4→CO2+2SO2+2H2O
Conc. H2SO4 oxidises C to produce two gaseous products.
New Question
10 months agoContributor-Level 10
The specialisations offered by Nehru College of Nursing, Panayur in the MSc programme are:
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (i)
Peroxy linkage refers to the presence of a bond between oxygen and oxygen (O-O) in a molecule.
Only H2S2O6 and H2S2O7 of the sulphur oxoacids above have a peroxy linkage.
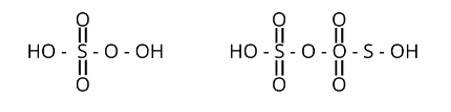
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
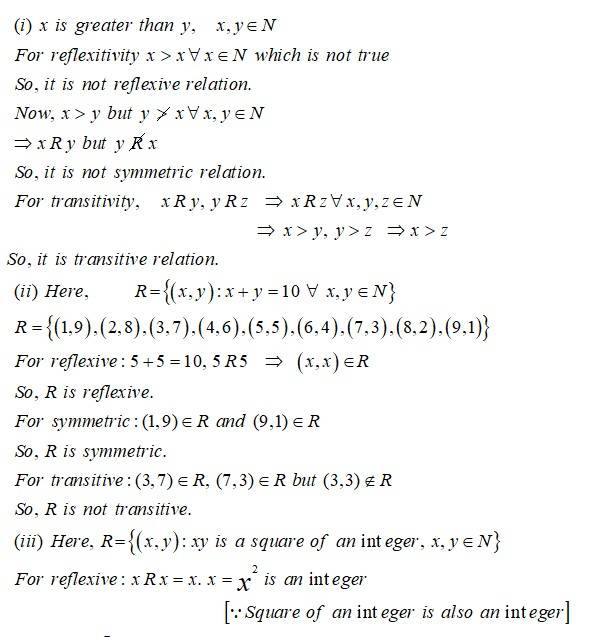

New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (iii)
The C-atom in the CO32- ion undergoes sp2 hybridization. BF4, NH4+ and SO42- have a tetrahedral structure, whereas it has a triangular planar structure.
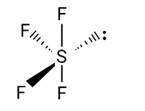
New Question
10 months agoContributor-Level 10
The course fee for the MSc programme at Nehru College of Nursing, Panayur is INR 2 lakh. The course fee is an amalgamation of various fee componenets such as the tuition fee, hostel fee, and the admission charges. Candidates are required to pay the course fee through the modes offered by the institute.
Candidates who get the admission offer are eligible to pay the course fee.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
New Question
10 months agoContributor-Level 10
As per popularity basis, listed below are the top Fashion Designing colleges in Noida along with their tuition fees:
| Top Colleges | Tuition Fee |
|---|---|
| Amity School of Fashion Technology | INR 3 Lacs - INR 12 lakh |
| IMS Design and Innovation Academy | INR 1 Lacs - INR 16 lakh |
| Amity University, Noida | INR 3 lakh |
| AAFT | INR 2 Lacs - INR 9 lakh |
| Satyam Fashion Institute | INR 7 lakh |
Disclaimer: This information is sourced from official website and may vary.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (iii)
Na (H2)PO2
1+ (2x+1)+ x +2 (−2)=0
x−1=0
x=1
New Question
10 months agoContributor-Level 10
There are about 15+ best Fashion Design colleges in Noida. Of these, 15 colleges are privately owned and 1 college is owned by public/government organisations. Students can secure admission after passing Class 12 boards. Some of the top Design colleges in Noida offer Fashion Design courses include Amity School of Fashion Technology, IMS Design and Innovation Academy, Amity University, Noida, AAFT, Satyam Fashion Institute (Only for Girls), Apparel Training and Design Centre, and many others.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
New Question
10 months agoContributor-Level 9
Job Profile | Average Salary |
|---|---|
Bank Probationary Officer (PO) | INR 5 LPA to INR 7 LPA |
LIC AAO | INR 3 LPA to INR 4 LPA |
IBPS Clerk | INR 2 LPA to INR 3 LPA |
RRB NTPC | INR 6 LPA to INR 8 LPA |
SSC CHSL | INR 3 LPA to INR 4 LPA |
IBPS PO | INR 5 LPA to INR 6 LPA |
Note: The above-mentioned are industry-based average payouts. The actual salary may differ from student to student based on their skills and competency.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (i)
The catalytic oxidation of ammonia produces NO gas, which is used to make HNO3. 4 moles of NH3 created 4 moles of NO in the equation below. As a result, the moles of NO produced by oxidising two moles of NH3 will be two moles.
4NH3 + 5O2→4NO + 6H2O.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
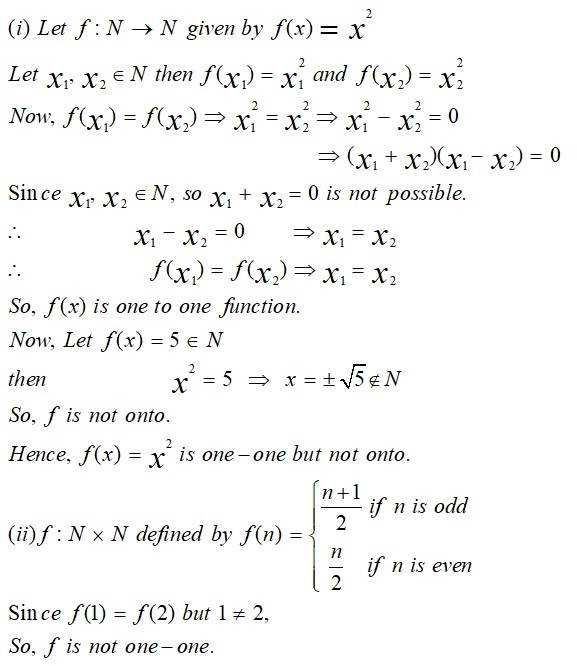
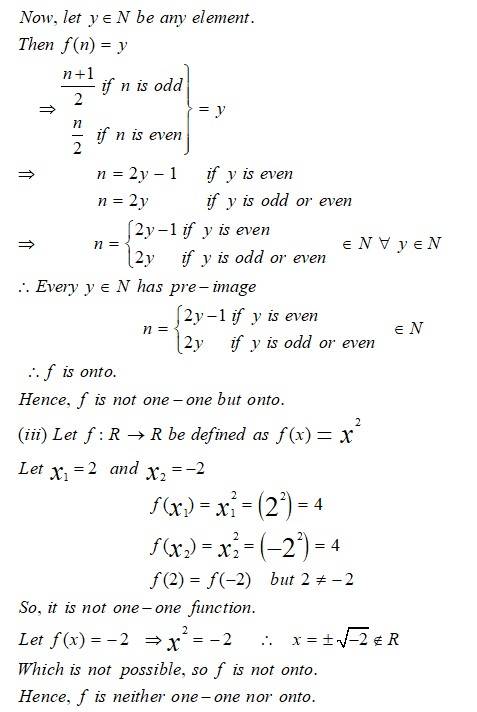
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (i)
We get N2 in both situations when we heat ammonium dichromate and barium azide separately.
(NH4)2Cr2O7→Cr2O3 + 4H2O + N2
Ba (N3)2→Ba + 3N2.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
