Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
Yes, SLS Pandit Deendayal Energy University registration window is currently open. The eligible students can visit the official website to register and apply for the BCom (H) courses. Admission criteria is entrance-based at the university. Moreover, the applications are accepted in an online mode by the university. Eligible students can visit the official website to register and apply.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(a, b, d) a) total mechanical energy of the body at any time t is
E= w2a2
KE=1/2mv2=
Kmax= ½ mw2a2=E
b) K= ½ mw2a2cos2wt
for a cycle value of coswt is =1/2
= 1/4 mw2a2= kmax/2
c) v=dx/dt = a coswt
Vmaen =Vmax+Vmin/2
= aw-aw/2=0
d)Vrms=
Vrms=Vmax/
New Question
10 months agoContributor-Level 10
For admission to the BSc course at Cheran College of Nursing, students need to fulfil the minimum eligibility criteria in the first place. The selection criteria for this course is merit-based and candidates must pass Class 12. The duration of the course is four years.
New Question
10 months agoContributor-Level 10
MAKAUT is affiliated with around 40 colleges; the total number of seats offered in the colleges and university is around 6,670. From which only two colleges offer the highest number of BSc seats. Check out the name of the college with the number of seats offered for the BSc course in the table below:
| Institutes | Seats |
|---|---|
| Institute of Management Studies | 570 |
| iLEAD Kolkata | 570 |
New Question
10 months agoContributor-Level 10
Many students wish to clear their board exams without studying, however, please note that you will not be able to pass the 12th class without securing the minimum passing marks of 33 in all subjects and also 33 percent in aggregate.
In case you are unable to score the passing marks in one or two subjects, you will be allowed to sit for the supplementary exams. You are advised to go through the Andhra Pradesh Inter 2nd year syllabus, and know the exam pattern so that you are able to build a strategy to score well in exams. Make notes of important points as you study. Stay confident and focus your energy on studies and you'll be able to p
New Question
10 months agoContributor-Level 10
Yes, BSc course is available at Cheran College of Nursing. The institute offers BSc courses at the UG level. Candidate must meet the eligibility criteria to enrol for BSc course admission set by the college. Aspirants must pass Class 12. Cheran College of Nursing's admission process is merit-based.
New Question
10 months agoContributor-Level 10
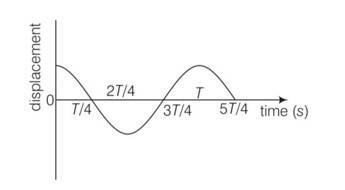
This is a multiple choice answer as classified in NCERT Exemplar
(a, b, c) At t= 3T/4, the displacement of the particle is zero. Hence particle executing SHM will be at mean position i.e x=0 acceleration is zero and force is also zero.
At t= 4T/3, displacement is maximum i.e extreme position, so acceleration is maximum
At t = T/4 corresponds to mean position, so velocity will be maximum at this position.
At t= T/2 corresponds to extreme position so KE =0 and PE =maximum.
New Question
10 months agoContributor-Level 7
With this subject combo, you're eligible for many Science and general courses. Although you cannot go for courses that strictly require Biology or Maths. But you still have good options like:
- B.Sc (Chemistry)
- B.Sc (Environmental Science)
- B.Sc (Food Science and Food Technology)
- B.Sc (Forensic Science) (some universities accept with Chemistry only)
- BA courses (like English Hons, General BA, etc.)
- B.Sc (Physical Science) (some colleges accept Chemistry without Maths)
Here are some good central or state universities where fees are reasonable:
- Delhi University — they offer many B.Sc and BA courses, very affordable (govt. fees around 10,
New Question
10 months agoContributor-Level 10
Yes, SLS, Pandit Deendayal Energy University offers BCom (Hons) course of four-year duration. It is available in a full-time mode of study. For admission, candidates have to take the entrance test of the school and selected students are called for the interview round. After this, the selected students can confirm their seats by paying the required fee amount.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(a, b, d) x=asinwt
V=dx/dt=awcoswt
A=dv/dt=-aw2sinwt
Force = mass = -mw2x
So force is directly proportional to displacement.
New Question
10 months agoContributor-Level 9
With 88 percentile in MBA CET and coming from general category it's unlikely to get an admission in PUMBA. PUMBA cutoff through CET for general category students is usually around 98 to 99 percentile.There is immense competition and all the seats are usually taken by top scorers. You can apply and get involved in the CAP rounds, because the cut off may differ slightly every year. You should also consider applying at other reputable colleges in Maharashtra accepting CET scores as there may be colleges with little lower cutoff. Stay ready with backup options.
New Question
10 months agoContributor-Level 10
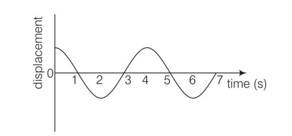
This is a multiple choice answer as classified in NCERT Exemplar
(b), (d) It is clear the curve that points corresponding to t=2s and t=6s are separated by a distance belongs to one time period. hence these points must be in same phase
Similarly points belongs t-1s to t=5sare at separation of one time period. Hence must be in phase.
New Question
10 months agoContributor-Level 10
MSc Nursing full form is Master of Science in Nursing, a two-year-long PG degree program, intended to provide advanced education and training for Registered Nurses. This course helps students develop strong clinical, teaching, and research skills, preparing them for nursing education, management, and leadership roles. Students can check the list of career options available after completing MSc from Cheran College of Nursing:
| Job Profiles | Job Description | Average Salary |
|---|---|---|
| Clinical Nurse Managers | Nurse managers supervise teams of nurses on the ground, whereas nurse leaders have a higher level, mainly administrative responsibility. | INR 6.5 LPA |
| Staff Nurses | Throughout the treatment procedure, staff nurses monitor the patient's physical and mental well-being. | INR 2.9 LPA |
| Nurse Educators | Nurse educators primarily instruct future nurses on clinical skills, patient care procedures, and best collaborative practices. | INR 4.9 LPA |
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(a), (c) consider the motion of a ball inside a smooth curved bowl
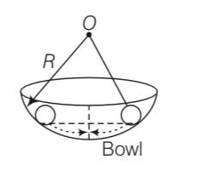
for small angular displacement or slightly released motion, it can be considered as angular SHM.
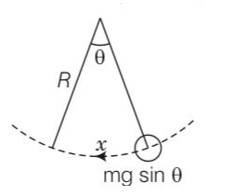
When the ball is at angle of the restoring force acts
Ma=mgsin
A= gsin
Hence motion is SHM
So T=
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: Correct option: D
[Fe (CN)6]3- ion shows magnetic moment corresponding to one unpaired electron. (CN) is a ligand with a strong field that couples electrons, resulting in hybridisation. d2sp3 and it has one unpaired electron, it has the magnetic moment of one paired electron.
μ= = 1 (1 + 2)
= 3 = 1.73BM
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(a), (c) the rotation of earth about its axis is periodic because it repeats after a regular interval of time.
The rotation of earth is obviously not a to and fro type of motion about a fixed point. So its motion is SHM.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: Correct option: B
From the Cis and Trans forms, geometrical isomerism forms compounds. All of the valences in the MX6 ligand are the same, resulting in an identical molecule. L is a distinct ligand in MX5L, and it can be replaced with any other valency in the structure to create an identical molecule; the molecule does not change. Because of the presence of symmetry, which is an essential prerequisite for revealing geometrical isomerism, complexes do not show geometrical isomerism.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: Correct option: A
The term "linkage isomerism" refers to two molecules that have similar ligands but differ in the donating site of the ligand. Ambidentate is made up of two different types of donor atoms.
Example [Fe (NO2) (H2O)5]Cl2. NO2 is Ambidentate ligand as Nand O2 are donors.
As a result, linkage isomerism occurs in coordinations with ambidentate ligands. Reason and Assertion are both correct.
New Question
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: Correct option: B
[Cr (H2O)6]Cl2 and [Fe (H2O)6]Cl2
Due to the creation of a more stable complex ion after obtaining ion, [Cr (H2O)6]Cl2 and [Fe (H2O)6]Cl2 are reduced in nature. Both compounds have a coordination of 6 and so form an octahedral complex. The hybridisation state of is 3 and the oxidation state of is 2+. Hybridization is 3d4 and 3d6 is 2+. Both compounds have unpaired electrons and have weak field ligands. In both compounds, removing electrons is simple.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts