Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
No, Indian Institute of Management Jammu does not accept direct admission. The institute accepts admission based on the entrance exam. IIM Jammu accepts the CAT (Common Admission Test) for MBA admission. After qualifying for the exam, students need to appear for the personal interview.
New Question
10 months agoContributor-Level 10
After completing your TN Class 12 in Science stream, you can appear for JEE Main, NEET, JEE Advanced, BITSAT, COMED-K, IPU-CET, AMU Entrance Exam, AIIMS BSc Nursing, AIIMS Paramedical and more. For detailed information on entrance exams after Class 12 Science, students can click on the link provided below:
New Question
10 months agoContributor-Level 7
The PTET counselling schedule will begin after the results are declared. The counselling will be conducted in multiple rounds to accommodate candidates with different ranks for the BEd course across participating colleges in Rajasthan. Candidates should regularly check the official website for updates regarding the exact schedule and important dates to avoid missing any deadlines. We will also share the detailed schedule as and when it is released.
New Question
10 months agoGuide-Level 15
DoMS IIT Roorkee offers admission in Executive MBA based on the merit of the aspirants in the last qualifying exam. The institute considers the applications submitted by the aspirant for selection. Further, candidates need to pass a personal interview round for admission. There is no entrance exam required to get into EMBA.
New Question
10 months agoContributor-Level 8
Lloyd Institute of Management and Technology (Pharm.) is ranked in the rank band of 101-125 under the Pharmacy category by the NIRF 2024 rankings. The Institute is a tier 2 institute offering courses such as BPharma, MPharma, and DPharma in full-time mode. The Institute offers respective courses in affiliation with AKTU, approved by AICTE and PCI, and accredited by NBA.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(d) y= asinwt + bcoswt
a=Asin and b= Acos
a2+b2=A2sin2 +A2cos2
A=
y=Asin +Acos
= Asin (wt+ )
dy/dt= Awcos (wt+ )
So it is proportional to displacement . so follows SHM
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: A and C
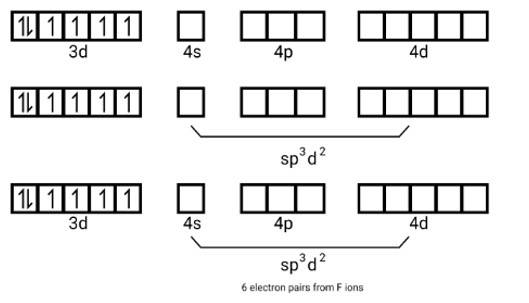
For answer at (A) Atomic number of Iron is 26 . In ferricyanide complex, iron is in oxidation state of 3+. The orbitals on the metal atom undergoes d2sp3 hybridizaion and hence the complex has octahedral shape.
Also it can be said that, [Fe (CN)6]3- has one unpaired electron which makes it weakiy paramagnetic. Therefore, option C is also correct.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(c) x= acoswt
Y= asinwt
Squaring and adding above eqns
x2+y2=a2, this is the equation of circle
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: A and C
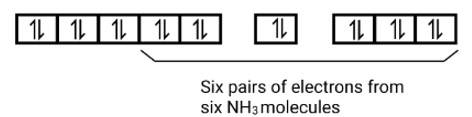
The outer octahedral complexes are weak ligands, number of pairing electrons and high spinning potential.
[MnCl6]3- - electronic configuration of Mn = 3d5,4s2. Shifting the valence electrons become
Mn3+ ,3d4 and number of unpaired electrons is 4.
[FeF6]3- - electronic configuration of Fe = 3d6,4s2. Shifting the valence electrons become
Fe3+ ,3d5 and the number of unpaired electrons is 5.
[CoF6]3 - electronic configuration of Co = 3d7,4s2 shifting the valence electrons become
Fe3+ ,3d6
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: A and C
(i) Orbitals of Co3+ ion d2sp3 hybridised orbitals of Co3+ [Co (NH3)6]3+ (inner orbital or low spin complex)
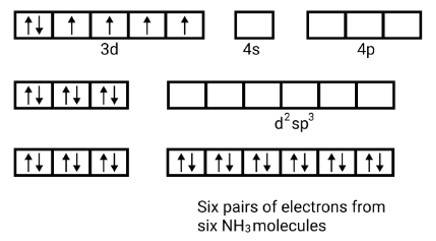
No. of unpaired electron =0
Magnetic property = diamagnetic, due to absence of unpaired electrons
(ii) Electronic configuration is 3 d6 orbitals of Fe2+ ion:
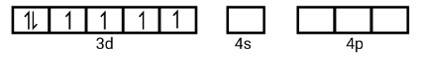

6 electron pairs from CN− ions occupy the s
New Question
10 months agoContributor-Level 10
Apart from the SICSR Pune MBA programme tuition fee, the total course fee consist of a range of fee components, including the admission fee, hostel fee, security charges, and some other miscellaneous charges. The following SICSR MBA fee breakup can be taken into consideration by the candidates:
| Fee components | Amount (for 2 years) |
|---|---|
INR 10.8 Lacs - INR 12.13 lakh | |
INR 4.74 lakh | |
INR 20,000 | |
INR 15.74 Lacs - INR 17.07 lakh |
NOTE: The mentioned fee is taken from the official website/ sanctioning body. It is still subject to change and, hence, is indicative.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(c) Consider the diagram in which a liquid column oscillates . in this case, restoring force acts on the liquid due to gravity.
Restoring force f = weight of liquid column of height 2y
t=-A = -2A
f
motion is SHM with force constant k= 2A
T= = 2
So time period is independent upon density of liquid.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: C
[Pt (NH3)2Cl (NO2)] is a neutral molecule. Ligands are arranged alphabetically.
So, NH3 Diammine, chloride and the embedded linkage is nitrogen and last is the metal name.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: B
Hydrate isomerism is isomerism in which water is used as a solvent.
Coordination compounds with the same composition but varying ligand connectivity are known as linkage isomers.
Solvate isomerism has the same composition as free solvent but distinct solvent ligand molecules.
Except for the ligand that swaps places with the anion, ionisation isomers are identical isomers.
Coordination isomers are coordination compounds with distinct metal and ligand compositions.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
To get admission to IIM Jammu where studnets generally need to clear the Common Entrance Test (CAT) and then participate in institute selection process. It includes the Personal Interview and Written Ability Test. The specific CAT percentile required for shortlisting may vary each year and by category. IIM Jammu prepares a merit list based on your performance in CAT, PI, WAT, and your overall profile (academic record, work experience, and diversity).
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: B
Ligands are neutral ions that form a coordination complex with the central metal atom.
NH4+ does not have a lone pair of electrons to give.
New Question
10 months agoContributor-Level 10
Yes, you can join B Tech through lateral entry admission. For this you have to do Polytechnic Diploma course from government authorized Polytechnic college. It is mandatory to pass 10th grade to join the 3 year Polytechnic Diploma course. Once you successfully complete Polytechnic course, you can appear for the Lateral entry entrance test and if you clear the entrance exam, you will get admission to second year of the B Tech course.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: A
A chelating agent binds to a single metal ion with two or more donor atoms. Oxaloto, Glycinato, and ethane-1,2-diamine all have two donor oxygen atoms. The gland Thiosulpato is ambidentate.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts