Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
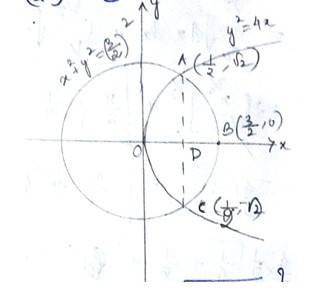
The given area of the circle is is a circle with centre (0,0) and radius, and the parabola is -------------(2)
Solving (1) and (2) for x and y.
For,
Which is not possible.
For,
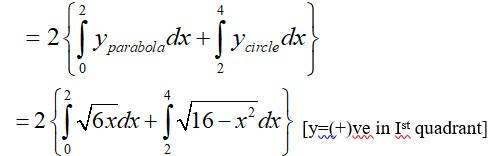

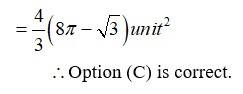
New Question
10 months agoContributor-Level 10
38. (C) Tm
In the periodic table, actinides lie from atomic number 90 to 103.
Thulium is not an actinide; it is a lanthanide.
New Question
10 months agoContributor-Level 10
The given curve is
Which is in the form of a parabola nad the lines are
At
At
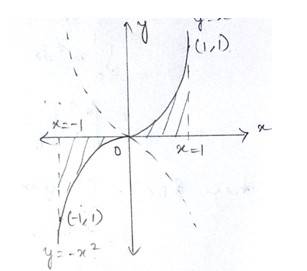
Shaded area of the Ist quadrant
Shaded area of the IInd quadrant
Total area of the enclosed region
Option (c) is correct.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
37. (D) Mn2+ acts as auto catalyst
Mn2+ formed in the reaction acts as an autocatalyst. Auto catalyst is a compound that is one of the products that are formed in the reaction itself catalyses the reaction.
Reaction: 2MnO4- + 16H+ + 5C2O42- → 2Mn2 + + 10CO2 + 8H2O
New Question
10 months agoContributor-Level 10
55. (ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
Explanation: Hydrometallurgy is used to extract copper from low-grade ore. Hydrometallurgy entails dissolving the ore in a suitable reagent and then precipitating it. In this method, more electropositive metal is used, allowing pure metal to be displaced.
New Question
10 months agoContributor-Level 10
UCEOU is a constituent of Osmania University. Thus, the institute is expected to follow the fee refund policy framed by the university. As per the university's policy, students who withdraw their admissions are entitled to a refund of fees. However, the amount of refund depends on the following policy:
| Point of time when withdrawal request is received | Refund Amount |
|---|---|
| 15 days or more before the last date of admission | 100% refund |
| Less than 15 days before the last date of admission | 90% refund |
| 15 days or less after the last date of admission | 80% refund |
| 30 days or less after the last date of admission | 50% refund |
| More than 30 days after the last date of admission | No refund |
New Question
10 months agoContributor-Level 10
36. (A)i, ii
Disproportionation/redox reaction is a reaction in which one compound is oxidised as well as reduced at the same time.
(A) Cu+ ? Cu2+ + Cu
( + 1) ( + 2) (0)
(B) 3MnO4- + 4H+ ? 2MnO4- + MnO2 + 2H2O
( + 6) ( + 7) ( + 4)
New Question
10 months agoNew Question
10 months agoContributor-Level 10
54. (ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
Explanation: The zone refining method is very useful for producing high-purity semiconductors and other metals, such as germanium.
New Question
10 months agoContributor-Level 10
35. (B)+3
Out of all +3 is the most common oxidation state for all the lanthanide elements.
New Question
10 months agoContributor-Level 10
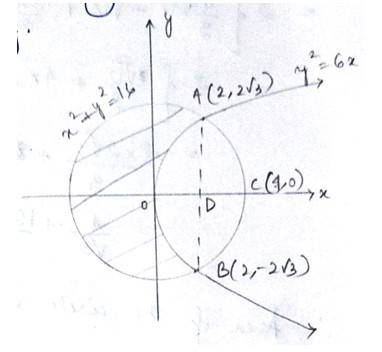
The given equation of curve i.e, - (1) is a parabola and
- (2) is a circle
With centre (0,0)and radius
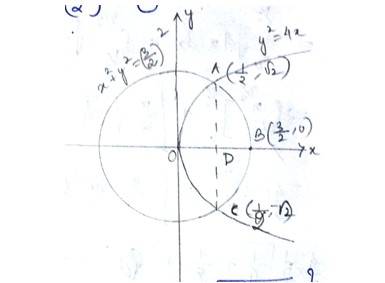
Solving (1) and (2) for x and y,

New Question
10 months agoContributor-Level 10
53. (ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
Explanation: Froth Flotation method is used to separate the hydrophobic materials from hydrophilic materials. In this method a mixture of palm oil, water and detergent is taken in a tank along with powdered sulfide ore. Compressed air is then passed through the pipe of the rotating agitator to create froth. The sulfide ore is then wetted by the palm oil mixture and it rises with the froth and the impurities or gauge settles at the bottom of the tank. The froth containing the sulfide is then cleaned and dried. Cresols or Aniline
New Question
10 months agoContributor-Level 10
Yes, there are many Textile Design colleges in India that offer certificate courses. Some of them are mentioned below along with their tuition fees:
Top Colleges | Tuition Fee |
|---|---|
NIFT Delhi | INR 1 Lacs – INR 2 lakh |
NIFT Bangalore | INR 1 lakh |
BHU | INR 5,000 |
New Delhi YMCA, Institute for Fashion Technology and Design | INR 29,000 – INR 54,000 |
Apparel Training and Design Centre | INR 25,500 |
Disclaimer: This information is sourced from the official website/ sanctioning body and may vary.
New Question
10 months agoContributor-Level 10
The given equation of the lines are
Area of

The point of intersection of the circle and the parabola is .
Taking in first quadrant
Area of
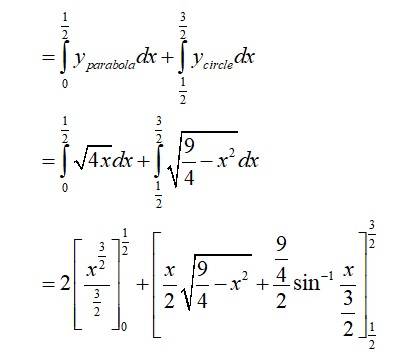
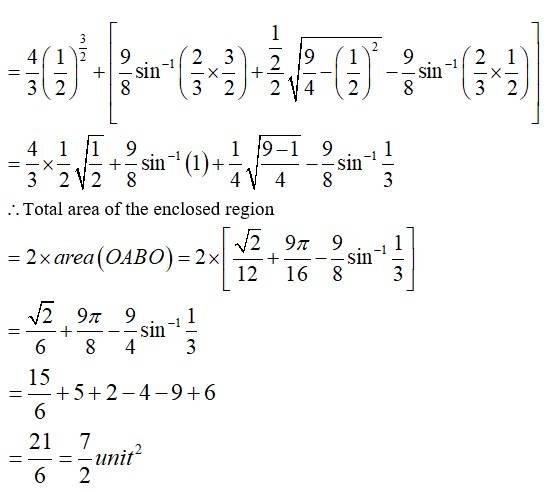
New Question
10 months agoContributor-Level 6
You can find the TS inter syllabus for 1st and 2nd year on the official website, tgbie.cgg.gov.in. On the home page, there is the syllabus tab on the left, click on it and you'll find subject-wise syllabus for TS Intermediate exams. However, the you'll have to find syllabus for different subjects there as they are provided in a simple format.
You can find the updated TS inter syllabus 2026 subject-wise by clicking on the link provided here. Click and download the syllabus and start preparing for your exams. The syllabus mentions all the topics that you need to prepare to score well in the exams.
New Question
10 months agoContributor-Level 10
Yes, there are many BDes in Textile Design colleges in India. Some of them are mentioned below along with their tuition fees:
Top Colleges | Tuition Fee |
|---|---|
NIFT Delhi | INR 12 lakh |
Amity University Mumbai | INR 5 lakh |
NIFT Bangalore | INR 6 Lacs – INR 12 lakh |
Pearl Academy Delhi | INR 16 lakh |
NIFT Mumbai | INR 12 lakh |
Disclaimer: This information is sourced from the official website/ sanctioning body and may vary.
New Question
10 months agoContributor-Level 10
52. (i) Both assertion and reason are true and reason is the correct explanation of assertion.
Explanation: Van Arkel method is generally used to obtain pure forms of Zirconium (Zr) and Titanium (Ti).
The metal iodide is heated at 1800 K and is decomposed on a tungsten filament. The pure metal is dropped on the tungsten filament. This proves that ZrI4 is volatile and decomposes at 1800 K.
New Question
10 months agoContributor-Level 10
34. (B) 3d5
Magnetic moment of an electron/dipole moment is caused by its intrinsic properties of spin and electric charge. It depends upon the number of unpaired electrons in its valence shell. The more the number of unpaired electrons, the greater will be the value of magnetic moment.
3d7 = 3 unpaired electrons
3d5 = 5 unpaired electrons
3d8 = 2 unpaired electrons
3d2 = 2 unpaired electrons
Out of all, 3d5 has five unpaired electrons that is maximum and hence it has the highest magnetic moment.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts