Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
47. Option (ii) A (4) B (3) C (1) D (2)
Coloured bands are found in chromatography.
Hence, option (A) from column I is matched with option (4) from column II. Impure metals are converted to volatile complexes in Mond's process.
Hence, option (B) from column I is matched with option (3) from column II. Purification of Ge and silicon is done using zone refining.
Hence, option (C) from column I is matched with option (1) from column II. Purification of mercury is done using fractional distillation.
Hence, option (D) from column I is matched with option (2) from column II.
New Question
10 months agoContributor-Level 10
For admission to the BTech course at Vikram Sarabhai Institute of Engineering & Technology, students need to fulfil the minimum eligibility criteria in the first place. The selection criteria for this course is entrance-based i.e., Entrance Exam (aptitude and suitability)/UPSEE Counselling and candidates must pass Class 12. The duration of the course is four years spread over eight semesters.
New Question
10 months agoContributor-Level 10
Students are advised to correct the errors even if it as a minor mistake. They must inform the school authorities about the error in the Karnataka 2nd PUC hall ticket. School authorities will help you in getting the new Karnataka 2nd PUC hall ticket without any mistakes.
New Question
10 months agoContributor-Level 10
The given equation of the curve is - (1) and
the line is - (2)
Solving (1) and (2) for x and y
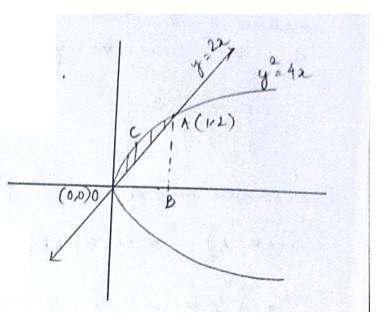
So,
for we get
for , we get
so, the point of intersection are (0,0)and (1,2)
area (DCAO)=area (DCABO)-area ( )
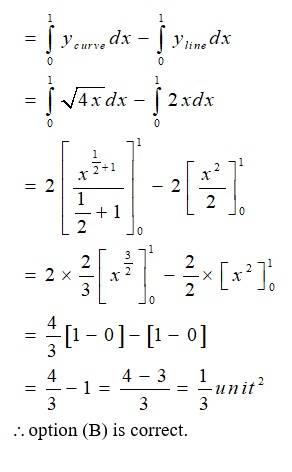
New Question
10 months agoContributor-Level 10
The eligibility and selection criteria for courses at SDIET are as follows:
| Courses | Selection Criteria | Eligibility |
|---|---|---|
BBA (3 courses) | Merit based | Class 12 with a minimum aggregate of 45% |
B.E. / B.Tech (3 courses) | JEE Main | Class 12 with a minimum eligibility of 60% |
BCA (2 courses) | Merit based | Class 12 with a minimum eligibility of 45% |
Candidates are required to meet the eligibility criteria mentioned above in order to apply successfully to the courses available.
New Question
10 months agoContributor-Level 10
46. Option (ii) A (2) B (4) C (5) D (3)
Pendulum is always made of nickel steel.
Hence, option (A) from column I is matched with option (2) from column II. Malachite is the ore of copper.
Hence, option (B) from column I is matched with option (4) from column II. Calamine is the ore of zinc.
Hence, option (C) from column I is matched with option (5) from column II. Cryolite is an ore of aluminum.
Hence, option (D) from column I is matched with option (3) from column II.
New Question
10 months agoContributor-Level 10
The UCEOU BE admission process begins with the application stage. In this stage, students are required to fill out the application form released by the institute Apart from this, students also have to register for TS EAMCET. The fee to apply for the exam counselling process ranges from INR 1,000 to INR 1,800.
Once the exam results are announced, eligible students must participate in the TS EAMCET counselling process. Students who are offered a seat are further required to report to the institute for verification of documents and payment of fees. A successful fee payment completes the admission process.
New Question
10 months agoContributor-Level 10
For BTech course admission, candidates must meet the eligibility criteria set by Vikram Sarabhai Institute of Engineering & Technology. Aspirants should pass class 12 with a minimum 45% aggregate. The admission criteria for the course is entrance-based. Bachelor of Technology, an undergraduate-level engineering degree course with a duration of four years. B Tech is a professional programme related to computers and technology that teaches students about the fundamentals of programming and computer software and hardware.
New Question
10 months agoContributor-Level 10
28. Ionisation enthalpies are the main factor that influence the reactivity of transition elements. Higher the ionisation enthalpy, lesser is the reacting of the transition element.
When we move along the period from Sc to Cu, a regular increase in the ionisation enthalpy is observed which results in the almost regular decrease in the reactivity of elements.
New Question
10 months agoContributor-Level 10
The equation of circle is which has centre at (0,0) & radius,
And the line
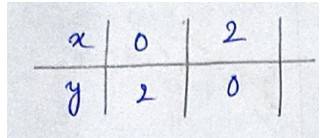
The smaller area of circle is given by
Area (ABCA) area (BOAB) – area (BOA)
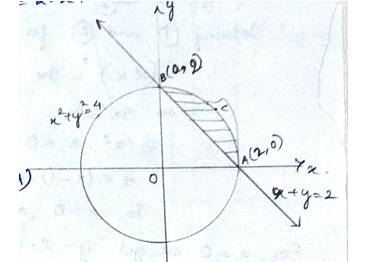

New Question
10 months agoContributor-Level 10
27. As per (n + l) rule, 4s has lower energy than 3d-orbital.
3d−n+l = 3+2 = 5
4s - n + l = 4 + 0 = 4
So, 4s are filled first.
After filling of electrons, 4s-orbital moves beyond 3d-orbital and 4s electrons are loosely held by the nucleus. Hence, electrons are removed first during the process of ionisation.
New Question
10 months agoContributor-Level 10
The given equation of the sides of triangle is
--------------------(1)
-------------------(2)
-------------------------(3)
Solving eqn (1) and (2) for x & y we get
The point of inersection of line (1)and (2)is A (0,1)
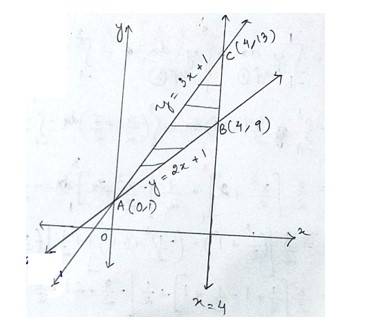
Putting x=4 in eq (1) and (2)we get,
The point of intersection of line (1)and (3) is B(4,9) and C (4,13)
Hence the required area enclosed ABC
New Question
10 months agoContributor-Level 10
The Riga Technical University and University of Malta are some of the top universities for international students in Northern Europe. Both the universities offer high placement rate, top-notch infrastructure, impressive ROI, best degree programs and more for its international students. The Riga Technical University placement rate is 95%, and the graduates are employed within 6 months of graduation. This figure indicates that out of 100 students, around 95 students secured employment. Whereas, University of Malta placement rate stands at 91%, and the graduates are employed within 6 months of graduation. This figure states that out of 10
New Question
10 months agoContributor-Level 10
To get into Vikram Sarabhai Institute of Engineering & Technology for BTech course, candidates must meet the eligibility criteria set by the institute. Students must pass Class 12 with a minimum of 45% aggregate (40% aggregate for ST/SC). VSIOET offers full-time BTech courses of four years' duration. Students can get into this course based on entrance-exam.
New Question
10 months agoContributor-Level 10
45. Option (ii) and (iii)
Explanation: Using oxidation method for extraction of chlorine from brine. The reactions involved are:
2Cl− + 2H2O → 2OH− + H2 + Cl2
For this reaction, the value of ΔG°=+422 kJ, which is positive. Using the formula ΔG°=−nE°F, we get a negative value of E° =−2.2 V.
New Question
10 months agoContributor-Level 10
26. As the positive charge of the ion increases or we can say that oxidation state of a transition element increases, its size decreases and as per Fajan's rule, more the charge on the metal ion, more is its tendency to form covalent compounds because positively charged cation attracts the electron cloud strongly towards itself.
New Question
10 months agoContributor-Level 10
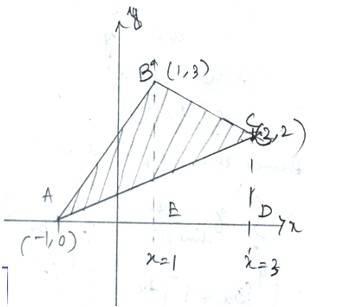
Let A (-1,0),B(1,3) and C (3,2) be the vertices of a triangle ABC
So, equation of line AB is
-------------(1)
Equation of line BC is
---------------(2)
Equation of line AC is
------------------------------(3)
Area of ABC= area ( )
New Question
10 months agoContributor-Level 10
Yes, the UCEOU BE courses are NIRF ranked. In the year 2022, the institute was ranked #117 by the NIRF Rankings under the 'Engineering' category.
New Question
10 months agoContributor-Level 10
There are generally five rounds in the NIFT Counselling. NIFT typically conducts multiple rounds of counselling depending on seat availability. If seats remain vacant after initial rounds, spot counselling may also be announced.
New Question
10 months agoContributor-Level 10
25. The sum of sublimation energy and ionisation enthalpy to oxidise cu (s) to Cu2+ is so highly that it is not compensated by the hydration enthalpy of Cu. Due to this, the Eof Cu is positive.
While in case if Zn, the E value is negative or more negative than the expected value because when the electrons are removed from the 4s-orbital. Zn acquires a stable 3d10 configuration state.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
