Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
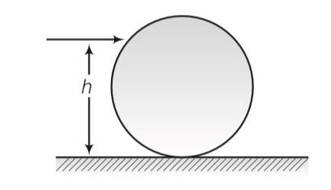
Consider a fig in which m is mass of sphere and R is the radius having h height above the floor
The sphere will roll without slipping when w=v/r where v is linear velocity and w is angular velocity .
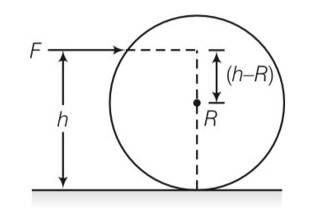
By conservation of momentum
mv (h-R)=Iw=2/5mR2 (v/R)
mv (h-R)=2/5mvR
h-R=2/5R
so h = 7/5R .d sphere will roll here so no loss of energy.
Torque = F (h-R)
For torque=0 h=R sphere will have only translational motion. It would lose energy by friction.
b
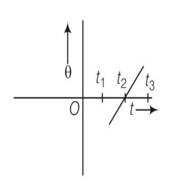
the sphere will spin clockwise when t>0 so h>R
so c and a
New Question
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
(i) When moving down the periodic table within a group, ionization enthalpy generally decreases due to increased atomic radius. However, in the case of gallium (Ga) and aluminum (Al), gallium experiences a higher effective nuclear charge due to less shielding from its inner electrons, resulting in a higher ionization enthalpy compared to aluminum.
(ii) As boron is smaller in size and the sum of its first three ionization enthalpies i.e. ΔH1+ΔH2+ΔH3 is very large so boron does not allow to lose its all three valence electrons and exist as
New Question
10 months agoContributor-Level 10
Yes, C Z Patel College of Business and Management offer scholarships to students as financial assistance. There are four types of scholarships available for students which are mentioned below:
| Scholarships | |
|---|---|
| Scholarship Name | Scholarship Details |
| 100% Scholarship | Scholarship is provided on the 1st Trimester Tuition fees. This will be awarded to students who secure the first rank in graduation at the university level without a backlog in any of semester. |
| 75% Scholarship | Scholarship on the 1st Trimester Tuition fees is awarded to students securing the second rank in graduation at the university level without a backlog in any of the semesters. |
| 50% Scholarship | 50% scholarship on the 1st Trimester Tuition fees is awarded to various students who secure third rank in the graduation at the university level without a backlog in any semester. |
| 10% of Tuition Fees | For students Charotar Vidya Mandal graduation programme not falling under any a, b, or c will get a scholarship of 10% of the tuition fees for the 1st Trimester. |
New Question
10 months agoContributor-Level 10
Candidates willing to get admission must take part in the C Z Patel College of Business and Management admission process before the last date of application. Candidates must know their C Z Patel College of Business and Management eligibility criteria and apply accordingly. Follow the C Z Patel College of Business and Management application process mentioned below:
Step 1: Visit the official website of the college.
Step 2: Select the required course.
Step 3: Pay the application fee.
Step 4: Fill out and submit the application form with the required information.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
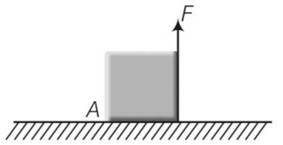
Moment of force 1= F (a)….anticlockwise
Moment of weight mg 2=mg (a/2)……. (clockwise)
Cube will not exhibit motion then 1 2
F=mg/2
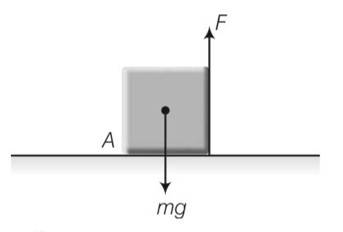
Cube will rotate when 1 2
F>mg/2
At a/3 from point A then
mg
when F=mg/4 which is less then mg/3 there will be no motion.
New Question
10 months agoContributor-Level 10
C Z Patel College of Business and Management offers PG and UG courses to students. These courses are taught to students across the Management and various other streams. At the UG level, the college offers BSc and BBA courses to students. At the PG level, the college offers MBA programmes to students. The MBA is available in three specialisations, namely Finance, Marketing, and Entrepreneurship. The following is the C Z Patel College of Business and Management seat intake:
Specialisation | Intake |
|---|---|
| Entrepreneurship | 60 |
| Finance | 60 |
| Marketing | 60 |
New Question
10 months agoContributor-Level 8
RPF Constable exam will be conducted by SSC. Both male and female candidates are eligible to apply for the exam. Candidates need to prepare well. Recruitment is done based on the vacancies.
New Question
10 months agoContributor-Level 10
Yes, applications for the Ramrao Adik Institute of Technology Certificate course can be filled out online. The institute provides an online application portal, which helps students apply from the convenience of their homes. In order to apply, students can visit the official website of the institute. After landing on the website, they are required to click on the preferred programme. After that, students must click on the register button, fill out the form displaying on the screen, and pay the application fees.
New Question
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
The following reaction takes place in the test tube
- i) When Aluminium is treated with dilute hydrochloric acid.
Al + HCl -à AlCl3
ii) When Aluminium is treated with dilute sodium hydroxide solution.
Al + NaOH -àNaAlO2 + H2
In both the cases, the Hydrogen atoms are evolved. So, when a burning match stick is brought near the test tube we hear a pop sound.
Aluminium does not react with concentrated nitric acid. A "layer" of "Aluminium Oxide" is formed, when aluminium reacts with the nitric acid because "nitric acid" is an oxidising
New Question
10 months agoContributor-Level 10
Hi, listed below are the top government BSc colleges in India with their tuition fee and eligibility criteria:
College Name | Tuition Fee | Eligibility / Exams |
|---|---|---|
INR 14,340 | Merit-Based |
Disclaimer: This information is sourced from official website and may vary.
New Question
10 months agoContributor-Level 10
Yes, C Z Patel College of Business and Management is one of the best choices for a student. C Z Patel College of Business and Management placements 2025 will be updated here once out. The C Z Patel College of Business and Management is visited by top companies for placements such as SOTC Travel, Marriott Surat, Hyatt Regency, and others. The highest package received by students is INR 16 LPA.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
As the slope of graph is positive and positive slope indicates anti clockwise rotation which is traditionally taken as positive.
New Question
10 months agoContributor-Level 10
Hi, listed below are the top private BSc colleges in Nagpur with their tuition fee and eligibility criteria:
College Name | Tuition Fee | Eligibility / Exams |
|---|---|---|
INR 3.91 lakh | Merit-Based | |
INR 34,650 – INR 54,400 | Merit-Based | |
G H Raisoni Institute of Engineering and Technology Admission | INR 1.44 lakh | Merit-Based |
INR 1.5 lakh | Merit-Based | |
INR 1.5 Lacs – INR 2.85 lakh | Merit-Based |
Disclaimer: This information is sourced from official website and may vary.
New Question
10 months agoContributor-Level 8
There is Physical Efficiency Test/Physical Measurement Test in RPF Constable exam. The structure of the exam is given below.
PET Standards
PET Standards | Male | Female |
|---|---|---|
1600 meters run | 5 min 45 secs | - |
800 meters run | - | 3 min 40 secs |
Long jump | 14 feet | 9 feet |
High jump | 4 feet | 3 feet |
PMT Standards:
Category | Height (in CMs) | Chest ( in CMs) ( Only for Male) | ||
|---|---|---|---|---|
Male | Female | Unexpanded | Expanded | |
UR/OBC | 165 | 157 | 80 | 85 |
SC/ST | 160 | 152 | 76.2 | 81.2 |
For Garhwalis, Gorkhas, Marathas, Dogras, Kumaonese and other Categories specified by Govt. | 163 | 155 | 80 | 85 |
New Question
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
BCl3+3H2O→B (OH)3+3HCl
B (OH)3+2H2O→ [B (OH)4]−+H3O+
B (OH)3 due to its incomplete octet accepts an electron pair (OH)- to give has configucation [B (OH)4]−.
Boron in this ion involves one 2s orbital and three 2p orbitals. Thus, hybridization of B in [B (OH)4]− is sp3
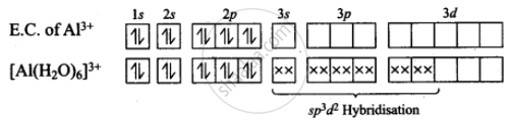
AlCl3 + 6H2O [Al (H2O)6]3+ +3Cl-
Hence, hybridization of Al is sp3d2
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The moment of inertia of the object I = r2 [sum of moment of inertia of each constituents particles]
All the mass in a cylinder lies at a distance R from the axis of symmetry but most of the mass of a solid sphere lies at a smaller distance than R.
New Question
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
When a trivalent atom is added to the crystal of SiO2, it substitutes silicon atoms which result in generation of holes. These holes make the crystal conductor of electricity. The overall Crystal is electrically neutral and is called the p-type conductor.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
When the vertical height of the objects is very small as compared to the earth's radius we call the objects small, otherwise it is extended. Building and ponds are small objects and deep lake and ocean are examples of extended objects.
New Question
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
As compared to carbon, silicon is bigger in size and is less electronegative. It shows resistance in forming p−p multiple bonding which is easily done by carbon. Thus, SiO2 is a 3−D network where each silicon is linked covalently to 4 oxygen atoms while in CO2, Carbon is linked with two oxygen atoms with double bond in a linear manner.
New Question
10 months agoContributor-Level 10
C Z Patel College of Business and Management popular programmes are BBA and BSc. BBA and BSc students are placed in top companies at top positions such as Hyatt Regency, Marriott, Vivanta By Taj, ITC Hotels, and others. BBA students can become Event Managers, or can join several other job profiles. The following are the USPs of joning the C Z Patel College of Business and Management:
- Industry Infused Mentorship for Students
- Immersive Learning Experience
- Accessible High-Quality Education, and others
Hence, choosing this college is the best decision.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts