Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
As we move down the group in group 13 and 14 the participation of s-electrons in bond formation decreases the primary reason behind this is the inert pair effect.
In this the p-electrons take part in bond formation and more energy is required to unpack the valence electrons to make them participate in bonding. Due to this the lower oxidation state of elements becomes stable done the hire oxidation state. As for group 13, +1 oxidation state is more stable than +3 and for group 14, +2 oxidation state is more stable than +4.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
Hi, the top specialisations offered by BSc colleges in Nagpur are:
| Top Specialisations | No. of Colleges |
|---|---|
| Computer Science | 10+ |
| Home Science | 10+ |
| Mathematics | 4 |
| Biotechnology | 4 |
| Chemistry | 4 |
| Forensic Science | 3 |
New Question
10 months agoContributor-Level 10
Yes, admissions at the C Z Patel College of Business and Management are quite wasy for hardworking students. Candidates must know their C Z Patel College of Business and Management eligibility criteria and apply accordingly. Follow the C Z Patel College of Business and Management application process mentioned below:
Step 1: Visit the official website of the college, which is czp.edu.in/.
Step 2: Select the required course.
Step 3: Pay the application fee.
Step 4: Fill out and submit the application form with the required information.
New Question
10 months agoContributor-Level 10
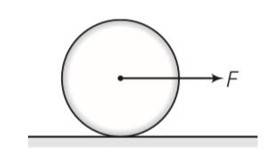
This is a long answer type question as classified in NCERT Exemplar
Frictional force f is acting in the opposite direction of F . let the acceleration of centre of mass of disc be a then
F-f=Ma where M is the mass of the disc
fR= (1/2 MR2)
so fR= (1/2MR2) (a/R)
Ma=2f
From the above equation
F = F/3
F< =
f/3 <
F=3
New Question
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
(A) Carbon has small size and large electronegativity, it forms strong n−pπ bonding with two oxygen atoms forming a separate CO2 molecule.
While in SiO2 silicon is larger in size with comparatively less electronegativity than carbon it shows no tendency to form n−pπ bonding rather forms Single covalent bond with oxygen. Thus, SiO2 possess 3D network-like structure in which each Silicon is bonded to 4 oxygen atoms.
(B) Carbon is smaller in size and lacks d-orbitals hence can have a maximum coordination number of four and sp3 hybridisation
New Question
10 months agoNew Question
10 months agoContributor-Level 10
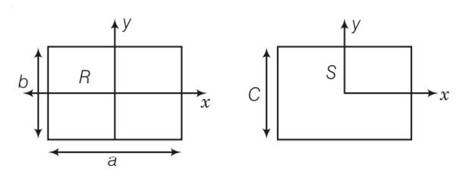
This is a long answer type question as classified in NCERT Exemplar
area of square = area of rectangular plate
C2=a b
(a)
here bxR
(b) here a>c so IyR>IyS
(c) IzR-IzS +2ab= (a-b)2
(IzR-IzS)>0
New Question
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
(A) sarbon in CCl4 does not have a vacant d-orbital to accommodate the electrons from OH of water molecules. Also CCl4 is nonpolar covalent compounds whereas H2O is polar. So, no strong interaction occurs between them. Hence CCl4 is miscible in water.
Whereas in SiCl4, silicon has bigger size than carbon and have d-orbitals for accommodation of electrons donated by OH of water in the process of hydroxylation. This leads to a strong interaction and silicon acid Is formed as a product. SiCl4 is completely miscible in water.
(B) As we move from carbon to sil
New Question
10 months agoContributor-Level 10
Hi, please refer to the table below for the highest Return on Investment for their students in the form of placement package:
College Name | Tuition Fee | Annual Median Package |
|---|---|---|
INR 14,340 | INR 3 LPA – IN 8.5 LPA | |
-/- | INR 3.48 LPA | |
INR 34,650 – INR 54,400 | INR 3.2 LPA | |
-/- | INR 3 LPA | |
-/- | INR 3 LPA |
Disclaimer: This information is sourced from official website/ media reports/ NIRF website and may vary.
New Question
10 months agoContributor-Level 10
C Z Patel College of Business and Management has a state-of-the-art infrastructure with multiple facilities available in the college. There are well-equipped training kitchens, housekeeping labs, front-office labs, and others. These facilities help students to learn the course without any trouble.
New Question
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
Both BCl3 and AlCl3 are electron deficient compounds that are central atom boron and aluminium have incomplete Octet. In each compound, a metal atom is surrounded by six electrons of three covalent bonds with 3 chlorine atoms.
Each chlorine atom has a complete Octet of eight electrons. The electron deficient compounds act as Lewis acid and readily accept two electrons to complete their octet.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
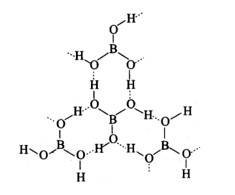
This is a Short Answers Type Questions as classified in NCERT Exemplar
H3BO3
Boric acid forms a hexagon and rings through hydrogen bonding and has a layer-like structure.
Boric acid is present in water as [B (OH)4]−.H3BO3 electron from the OH of water and forms the complex BOH for negative for sp3 and is present in sp3 hybridisation.
Reaction:
B (OH)3+2H2O→ [B (OH)4]− + H3O+
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
(a) Situation as given below
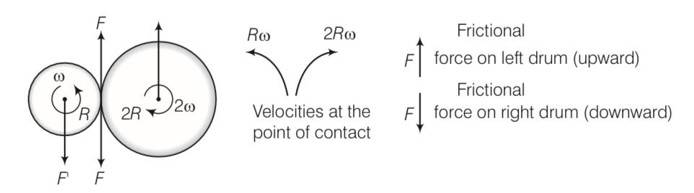
(b) F’ =F=F’’ where F’ and F’’ are external forces through support.
So Fnet=O
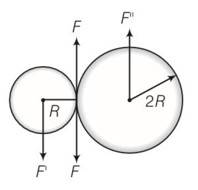
External torque = F (anticlockwise)
(c) Let w1 and w2 be the final angular velocities of smaller and bigger drum in both clockwise and anticlockwise . finally there will be no friction
hence Rw1=2Rw2
so
New Question
10 months agoNew Question
10 months agoContributor-Level 10
The fee range for colleges in India that offer an MBA in Business Analytics is given in the table below:
| Course Fees Range | No. of Colleges | College Names |
|---|---|---|
Less than INR 1 Lakh | 20+ colleges | Delhi School of Economics (DSE), SRM Easwari Engineering College, SNS College of Technology, S.A. Engineering College, etc. |
| INR 1-2 Lakh | 50+ colleges | NIT Trichy, Chandigarh Group of Colleges, Rajagiri Centre for Business Studies, Prestige Institute of Management and Research, etc. |
| INR 2-3 Lakh | 40+ colleges | MNIT Jaipur, Hindusthan College of Engineering and Technology, University of Engineering and Management, Apeejay Institute of Management and Engineering, etc. |
| INR 3-5 Lakh | 100+ colleges | IIT Dhanbad, Galgotias University, Chandigarh Group of Colleges, GLA University, Hindustan Institute of Technology and Science, etc. |
| More than INR 5 Lakh | 300+ colleges | IIM Bangalore, IIM Calcutta, Indian Institute of Foreign Trade, Management Development Institute, NMIMS, etc. |
Disclaimer: This information is sourced from the official website and may vary.
New Question
10 months agoContributor-Level 8
Yes, female candidates can apply for RPF Constable post. The age of the candidates should be between 18 years to 28 years. The educational qualification prescribed for the exam is Class 10 pass or equivalent from recognised board. Female candidates should have a minimum height of 157 cm. For both males and females, the chest measurement should be 80 cm (85 cm with expansion) for male candidates.
New Question
10 months agoNew Question
10 months agoContributor-Level 7
Those who are from Arts background and looking to specialise in Hotel Management field can opt for this degree. The course also helps in developing skills like customer service, communication, leadership and problem solving.
Hospitality management course usually involves working with people from various backgrounds and cultures, fostering a global perspective and cultural awareness.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts