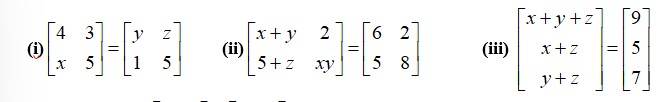Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
48. Option (ii) is correct since the arrow shows the direction of electron transfer. In the given reaction, the electron flows from π bond towards the electron-deficient H+ group . This can be represented as-

New Question
10 months agoContributor-Level 10
The DLIS MLISc application process is conducted online. Candidates willing to secure admission to the MLISc course offered at the department must follow a series of steps to compete the application process. Before applying, they must check the eligibility criteria laid down by the department. Eligible candidates can follow the steps listed below:
- Visit the official admission portal of Somaiya Vidyavihar University (SVU).
- Click on 'Register' and fill out the registration form.
- Log in to fill out the pre-admission form with all the required details.
- Submit a non-refundable application fee of INR 250 through the online payment gateway.
New Question
10 months agoContributor-Level 10
After being shortlisted for BSc at MSCAS, students must pay the full course fees to confirm their admission. This fee covers tuition, examination charges, and other administrative costs. Timely payment is essential to secure your seat and avoid losing admission to other students.
New Question
10 months agoContributor-Level 10
Motherhood University BA registrations may open around May/ June. The applications can be released following the Class 12 result announcement as well. The important dates may get published on the official website. Aspirants shall ensure that they submit their application forms on or before the prescribed deadline.
New Question
10 months agoContributor-Level 10
The MSCAS BSc selection process primarily considers students' Class 12 examination results, focusing on their academic performance in relevant subjects. The college prepares a merit list based on eligibility criteria specific to each specialisation. Shortlisted candidates are informed through the official website or via the contact details provided during registration.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
47. Bromine is found to be more electronegative than Carbon which leads to heterolytic fission. Due to the higher electronegativity the electrons displace from carbon to Br. Therefore, CH3 gets positive charge and Br gets negative charge and hence option (ii) is found to be the correct one.
New Question
10 months agoContributor-Level 10
To apply for the BSc course, you need to visit the official MSCAS application portal and complete the registration by providing your personal details. After verifying your email, you can log in to fill out the application form and pay the application fee of INR 500 online. Once submitted, keep a copy of your application for future reference.
New Question
10 months agoContributor-Level 10
As per the official website of Sai Nath University, there are no admission dates available for the BA admission. However, candidates can expect the BA admission dates to be out soon, since the admission portal is open for the 2025 admission. For admission to the BA courses, candidates must secure a valid score in the last qualifying exam.
New Question
10 months agoContributor-Level 10
(5) Equating the corresponding elements of the matrices we get,
a - b = -1 - (i)
2a + c = 5 - (2)
2a - b = 0 - (3)
3c + d = 13 - (4)
Subtracting eqn (1) from (3) we get,
2a - b (a - b) = 0- (-1)
2a - b - a+ b = 0 + 1 = 1
[a= 1]
Put we get,
2 × 1 + c = 5 c = 5 - 2 [c = 3]
Put (4) we get,
3 × 3 + d = 13 => d = 13 - 9 [d = 4.]
put we get, 1 - b = -1 b = 1 + 1 [b= 2]
New Question
10 months agoContributor-Level 10
(i)
corresponding
By equating the elements of the matrices, we get,
x= 1
y= 4
z= 3.
(ii)
By equating the corresponding elements of the matrices we get,
x+ y = 6 (I)
5 + Z = 5
xy = 8
x
putting eq in (1) we get
+ y = 6.
8 + y2 = 6y
y2 6y + 8 = 0.
y2 - 4y - 2y + 8 = 0
y (y-4) -2 (y-4) = 0
(y-4) (y-2) = 0
y= 4 0r y = 2.
When y = 4,x= 6-y = 6-4 = and z = 0.
Wheny = 2,x = 6-y = 6-2 = 4 and z = 0.
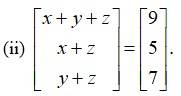
By equating the corresponding elements of the matrices we get,
x+ y + z = 9 -------(i)
x + z = 7 --------(ii)
y + z = 7 -------(iii)
Subtracting eqn (3) from (1) and (2) from (1) we get,
x + y + z -y - z = 9 - 7 and x
New Question
10 months agoContributor-Level 10
The course fees for three years BA in Sai Nath University range from INR 60,000 to INR 2.2 lakh. The course fee includes multiple other fee components, including one-time fee payment, others univeresity fees, and, more. Candidates must secure by depositing the required amount by the specified date. Failure to do so will result in the seats being allocated to the next the highest-ranking candidates. The Sai Nath University course fee may include additional components aside from tuition fees.
New Question
10 months agoContributor-Level 10
(E) (i) aij = such that i = 1, 2, 3 and j = 1, 2, 3, 4 for 3 × 4 matrix
So, a11= .
a12 =
a14 =
a21 =
a22 =
a23 =

New Question
10 months agoContributor-Level 10
46. This is a Multiple Choice Questions as classified in NCERT Exemplar
Carbocation is correct since H+ attacks on propene and the delocalization of electrons can take place in two possible ways which can be shown as-

New Question
10 months agoContributor-Level 10
(E) (i) aij such that i = 1, 2 and j = 1 * 2 for 2 * 2 matrix
Therefore a11 = A 2*2 =
a12 =
a21
a22 =
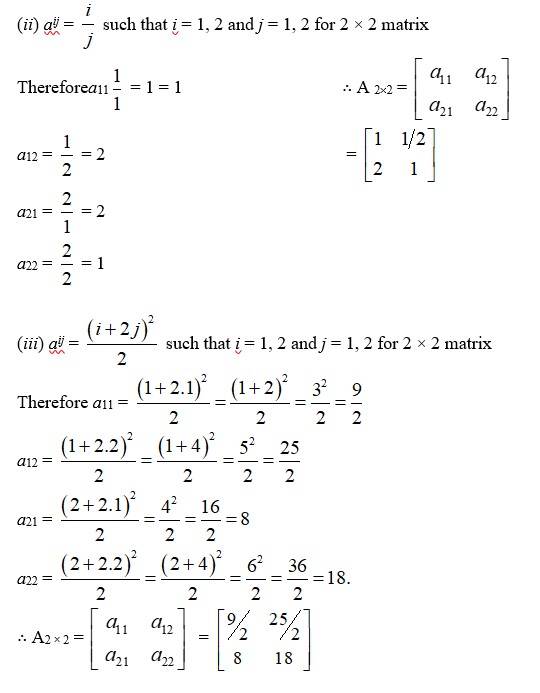
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
45. Option (iv) is found to be the correct one since the stabilisation of carboxylate ion depends upon the dispersal of the negative charge. And the dispersal depends upon two factors, i.e., +R effect of the carboxylate ion and Inductive effect of the halogens. In all the above structures, +R effect is common, but halogen atoms are uncommon. Therefore, dispersal of negative charge depends upon the halogen atoms. Fluorine is most electronegative, in structure (iv) two F atoms are present and hence more dispersal of negative charge is there.
New Question
10 months ago3.If a matrix has 18 elements, what are the possible orders it can have? What, if it has 5 elements?
Contributor-Level 10
As number of elements of matrix with order m * n
(E) Possible order of matrix with 18 elements are (1 * 18), (2 * 9), (3 * 6), (6 * 3), (9 * 2) and (18 * 1)
Similarly, possible order of matrix with 5 elements are (1 * 5) and (5 * 1)
New Question
10 months agoContributor-Level 10
Yes, Motherhood University offers a three-year-long BA programme. Additionally, it also provides integrated BA courses - BA B.Ed and BA LLB. Admission to the UG-level courses is granted on a merit-basis. Hence, the aspirants performance in Class 12 or equivalent exam is evaluated. Eligible students can apply for admission in both online and offline modes.
New Question
10 months agoContributor-Level 10
As, number of elements of matrix having order m * n = m.n.
(b) So, (possible) order of matrix with 24 elements are (1 * 24), (2 * 12), (3 * 8), (4 * 6), (6 * 4), (8 * 3), (12 * 2), 24 * 1).
Similarly, possible order of matrix with 13 elements are (1 * 13) and (13 * 1)
New Question
10 months agoRegister to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts