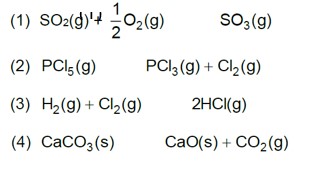
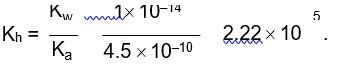7.30. At 473 K, the equilibrium constant Kc for the decomposition of phosphorus pentachloride (PCl5) is 8.3 x 10-3. If decomposition proceeds as:
PCl5 (g) ⇌ PCl3 (g) + Cl2 (g) ΔrHϴ = 124.0 kJ mol–1
(a) Write an expression for Kc for the reaction
(b) What is the value of Kc for the reverse reaction at the same temperature.
(c) What would be the effect on Kc if
(i) More of PCl5is added (ii) pressure is increased (iii) the temperature is increased.
7.30. At 473 K, the equilibrium constant Kc for the decomposition of phosphorus pentachloride (PCl5) is 8.3 x 10-3. If decomposition proceeds as:
PCl5 (g) ⇌ PCl3 (g) + Cl2 (g) ΔrHϴ = 124.0 kJ mol–1
(a) Write an expression for Kc for the reaction
(b) What is the value of Kc for the reverse reaction at the same temperature.
(c) What would be the effect on Kc if
(i) More of PCl5is added (ii) pressure is increased (iii) the temperature is increased.
(a) The equilibrium constant expression is
Kc = [PCl3] [Cl2] / [PCl5]
(b) The equilibrium constant expression for the reverse reaction is
Kc (reverse) = 1/ (8.3*103) =120.48
(c) (i) By adding more of PCl5, value of Kc will remain constant because there is no change in temperature.
(ii) When pressure
Similar Questions for you
0.01 M NaOH,
M = 1 * 10-2
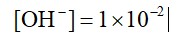
pOH = 2
pH = 2
Kp = Kc (RT)Dng
36 * 10–2 = Kc (0.0821 * 300)–1
Kc = 0.36 * 0.0821 * 300 = 8.86 » 9
A(g) ->B(g) + (g)
Initial moles n 0 &nbs
On increasing pressure, equilibrium moves in that direction where number of gaseous moles decreases.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 11th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering