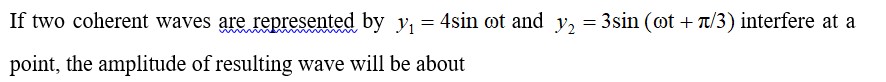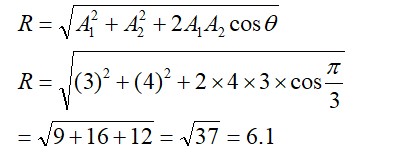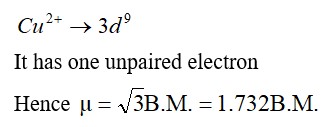8.28 Predict which of the following will be coloured in aqueous solution? Ti3+, V3+ , Cu+ , Sc 3+, Mn2+, Fe 3+ and Co 2+. Give reasons for each.
8.28 Predict which of the following will be coloured in aqueous solution? Ti3+, V3+ , Cu+ , Sc 3+, Mn2+, Fe 3+ and Co 2+. Give reasons for each.
8.28 Metal ions which have valence electrons in d-orbital and in which d-d transition can take place will be coloured and the metal ions which have completely filled orbital or have d- orbital will be colourless as no d-d transition is possible in those configurations.
Element | Atomic Number | Ionic State | Electronic configuration in ionic state |
Ti | 22 | Ti3+ | [Ar] 3d1 |
V | 23 | V3+ | [Ar] 3d2 |
Cu | 29 | Cu+ | [Ar] 3d10 |
Sc | 21 | Sc3+ | [Ar] |
Mn | 25 | Mn2+ | [Ar] 3d5 |
Fe | 26 | Fe3+ | [Ar] 3d5 |
Co | 27 | Co2+ | [Ar] 3d7 |
From the above table, it can be
Similar Questions for you
K2Cr2O7 + H2O2 + H2SO4->
Potassium permanganate in alkaline medium oxidise lodide to lodate.
Compound A is
KMnO4 decomposes upon heating at 513 K and forms K2MnO4 and MnO2.
2KMnO4
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering