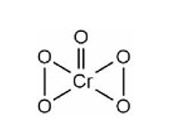
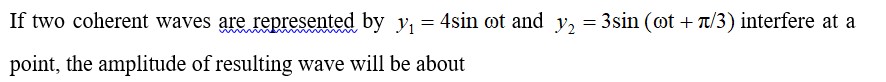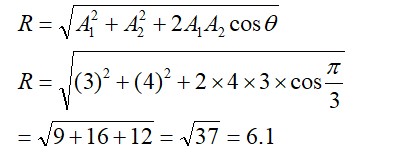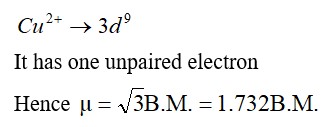8.29 Compare the stability of +2 oxidation state for the elements of the first transition series.
8.29 Compare the stability of +2 oxidation state for the elements of the first transition series.
8.29 It can be observed from the above table that in the starting of 3d transition series elements like Sc, Ti, V, Cr in +2 state are not that stable in their elements in the +3 state.
In the middle Mn2+, Fe2+, Co2+are quite known. In fact, Mn2+ and Mn7+ are most stable states in Mn. Fe2+ is less sta
Similar Questions for you
K2Cr2O7 + H2O2 + H2SO4->
Potassium permanganate in alkaline medium oxidise lodide to lodate.
Compound A is
KMnO4 decomposes upon heating at 513 K and forms K2MnO4 and MnO2.
2KMnO4
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry Ncert Solutions Class 12th 2023
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering