According to de Broglie, matter should exhibit dual behaviour, that is both particle and wave like properties. However, a cricket ball of mass 100 g does not move like a wave when it is thrown by a bowler at a speed of 100 km/h. Calculate the wavelength of the ball and explain why it does not show wave nature.
According to de Broglie, matter should exhibit dual behaviour, that is both particle and wave like properties. However, a cricket ball of mass 100 g does not move like a wave when it is thrown by a bowler at a speed of 100 km/h. Calculate the wavelength of the ball and explain why it does not show wave nature.
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: As per de Broglie, every object in motion has a wave character i.e. every object/matter have dual nature both particle and wave nature. But the wavelengths associated with ordinary objects are so short (because of their large
Similar Questions for you
Kindly go through the answers
(7.00)
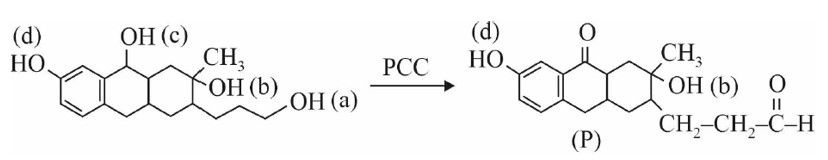
Kindly consider the following Image
In 4d orbital, n = 4 and
Radial nodes =
Radial nodes = 4 – 2 – 1 = 1
And angular nodes,
Here, number of unpaired electrons, n = 1
Spin only moment ;
= 173 × 10-2 B.M
=
= (At constant pressure)
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry NCERT Exemplar Solutions Class 11th Chapter Two 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering