The effect of uncertainty principle is significant only for motion of microscopic particles and is negligible for the macroscopic particles. Justify the statement with the help of a suitable example.
The effect of uncertainty principle is significant only for motion of microscopic particles and is negligible for the macroscopic particles. Justify the statement with the help of a suitable example.
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The uncertainty principle is significantly only for the microscopic particles and not for the macroscopic particles can be concluded by considering the following example. Let us consider a particle or an object of mass 1 milli
Similar Questions for you
Kindly go through the answers
(7.00)
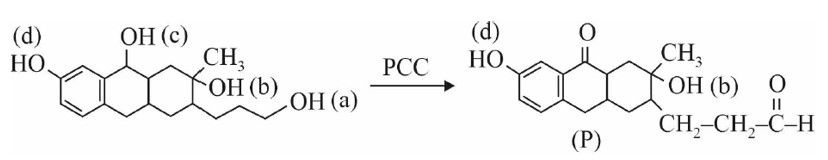
Kindly consider the following Image
In 4d orbital, n = 4 and
Radial nodes =
Radial nodes = 4 – 2 – 1 = 1
And angular nodes,
Here, number of unpaired electrons, n = 1
Spin only moment ;
= 173 × 10-2 B.M
=
= (At constant pressure)
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...

Chemistry NCERT Exemplar Solutions Class 11th Chapter Two 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering