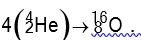Calculate the time interval between 33% decay and 67% decay if half-life of a substance is 20 minutes.
Calculate the time interval between 33% decay and 67% decay if half-life of a substance is 20 minutes.
Option 1 - <p>13 minutes</p>
Option 2 - <p>60 minutes<br><!-- [if !supportLineBreakNewLine]--><br><!--[endif]--></p>
Option 3 - <p>40 minutes<br><!-- [if !supportLineBreakNewLine]--><br><!--[endif]--></p>
Option 4 - <p>20 minutes</p>
2 Views|Posted 7 months ago
Asked by Shiksha User
1 Answer
A
Answered by
7 months ago
Correct Option - 4
Detailed Solution:
N? /N? = e?
For 33% decay, N? /N? = 0.67 ≈ 2/3.
2/3 = e? ⇒ t? = (1/λ)ln (3/2)
For 67% decay, N? /N? = 0.33 ≈ 1/3.
1/3 = e? ⇒ t? = (1/λ)ln (3)
Δt = t? - t? = (1/λ) [ln (3) - ln (3/2)] = (1/λ)ln (2) = T? /? = 20 min
Similar Questions for you
Q = [4 *4.0026 – 15.9994] *931.5 MeV
Q = 10.2 MeV
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
66K
Colleges
|
1.2K
Exams
|
6.9L
Reviews
|
1.9M
Answers
Learn more about...

Physics Nuclei 2025
View Exam DetailsMost viewed information
SummaryDidn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
or
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
or
See what others like you are asking & answering