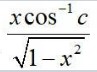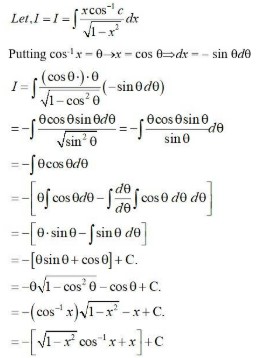Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 7
You must hold BA/BSC from any recognised university by UGC in India. And have atleast 55% and then CUET exam with good marks then you can
get admission easily.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
A Police Clearance Certificate (PCC) is an official document issued by Indian police authorities that confirms you have no criminal record. It is required by students who are applying for long-term visas to study abroad.
New Question
10 months agoContributor-Level 6
The Commission conducted the UPSC IES 2025 Prelims exam in an offline mode. It was held on June 8, 2025 at various centres across the country.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Radius = 1Ao
Volume of hydrogen molecules = 4/3 r3
= 4/3 (3.14) (10-10)3 m3
Number of moles of H2 = mass/molecular mass=0.5/2=0.25
Molecules of H2 present = number of moles of H2 present
= 0.25
So volume of molecules present = molecule number volume of each molecules
= 0.25
6 3
PiVi= PfVf
Vf = i= 3
Vf= 2.7 3
New Question
10 months agoNew Question
10 months agoBeginner-Level 5
After downloading the UP CPET 2025 admit card, applicants are advised to check the details mentioned in it. In case of any discrepancies in the admit card, students are advised to contact the exam conducting authorities via phone or through email ID and get the correction done in the details.
New Question
10 months agoContributor-Level 10
Some the top recruiters for Sasmira's Institute of Management Studies and Research are listed below:
Sasmira's Institute of Management Studies and Research Top recruiters | ||
|---|---|---|
Amazon | ITC | SBI |
Tata | Morgan Stanley | HDFC |
New Question
10 months agoContributor-Level 10
The institution has not released the average package details as of yet. Meanwhile, check out the highest and median packages offered at Sasmira's Institute of Management Studies and Research from table below:
Particulars | Placement Statistics (2025) |
|---|---|
the highest Package | INR 24 LPA |
Median Package | INR 7 LPA |
New Question
10 months agoContributor-Level 10
Admission to the BBA programme at DTU is subject to a merit list drawn on the basis of scores obtained by the students in best of four subjects of Class 12. Students, however, must have completed their Class 12 with 50% aggregate (with English as a subject) to be eligible for the course. Additionally, those appearing in the final exams of their Class 12 can also apply provided they can submit the proof of eligibility as per the date set by the university. Hence, yes, it is possible to get into DTU with 85% in Class 12 provided a student is shortlisted while creation of the merit list.
New Question
10 months agoContributor-Level 10
The Sasmira's Institute of Management Studies and Research provides good placements to its MBA graduating students. The students are given excellent placement support every year placing them in top companies and it is totally worth it. Check out the table below to know the key highlights of the Sasmira's Institute of Management Studies and Research for MBA in 2025:
Particulars | Placement Statistics (2025) |
|---|---|
the highest Package | INR 24 LPA |
Median Package | INR 7 LPA |
Top Recruiters | Amazon, SBI, ITC, etc. |
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
(a) The average KE will be the same
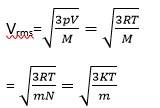
M= molar mass of the gas
m=mass of each molecular of the gas
R= gas constant
vrms
(b) k = Boltzmann constant
T= absolute temperature
mA>mB>mC
Vrms.A
New Question
10 months agoContributor-Level 6
A gerund is a verb form that ends in -ing but functions as a noun in a sentence. Although a gerund looks like a verb, it acts as a subject, object, or complement.
Example:
- Joey is taking acting classes.
- Monica is working as a chef at a restaurant.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts