Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
Ans.1.26
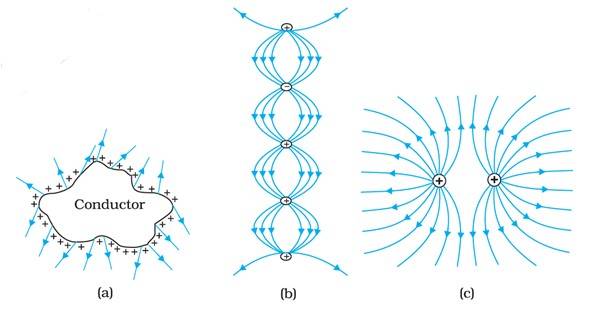
(a) The field lines showed in (a) do not represent electrostatic field lines because field lines must be normal to the surface of the conductor.
(b) The field lines shown in (b) do not represent electrostatic field lines because field lines can not emerge from a negative charge and cannot terminate at a positive charge.
(c) The field lines shown in (c) represent electrostatic field line. This is because the field lines emerge from the positive charge and repel each other.
(d) The field lines shown in (d) do not represent electrostatic field lines because the field lines should not intersect each other.
New Question
11 months agoNew Question
11 months agoNew Question
11 months agoContributor-Level 10
Several MNC's participated in the CMR Institute of Technology placements. The company-wise packages offered at CMRIT placement in 2025 are given below:
Particulars (Company) | Placement Statistics (2025) |
|---|---|
Belc | INR 44 LPA |
Hitachi Construction Plant | INR 36.7 LPA |
ARK | INR 33.62 LPA |
Kawashima Packaging Machinery Ltd. | INR 29.44 LPA |
PhonePe | INR 16 LPA |
Thermo Fisher Scientific | INR 12.95 LPA |
Amadeus | INR 12.62 LPA |
Maersk | INR 10.83 LPA |
Eurofins | INR 10.75 LPA |
Outcomes Operating India Pvt. Ltd. | INR 10.08 LPA |
Oracle CSS GSC | INR 9.34 LPA |
New Question
11 months agoContributor-Level 10
(d) BaCO3
Thermal stability is directly proportional to the size of the cation i.e., larger the size of the atom, greater is its thermal stability. Ba being the biggest cation among the given compounds, has a greater thermal stability.
New Question
11 months agoContributor-Level 10
Comparing BA course fees at DSMNRU and Maharishi University of Information Technology, DSMNRU is more affordable. DSMNRU offers a BA fee of INR 3,000, which is lower compared to the INR 1.2 lakh BA fee at Maharishi University of Information Technology. However, it must be noted that the mentioned fees have been taken from the official website and are still subject to change.
New Question
11 months agoContributor-Level 10
Doon University offers the BSc course in differet specialisations. The students looking forward to study BSc in this university must acquire the information about the fee structure as it is a important factor while selecting college. In Doon University the cost of studying the BSc course ranges from INR 60,000 to INR 1 lakh.
New Question
11 months agoContributor-Level 10
(a) Li
Li+ is the smallest in size and thus, has the highest charge density and hence attracts the water molecules more strongly to form hydrated salts.
New Question
11 months agoContributor-Level 10
The institute has not released the BTech-course wise average package as of yet. The CMRIT overall average package offered during the 2025 placements stood at INR 6.99 LPA. For a detailed analysis of CMR Institute of Technology average package trends refer to the table below:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
Average Package | INR 5.98 LPA | INR 6.26 LPA | INR 6.99 LPA |
New Question
11 months agoContributor-Level 10
No, Woxsen University PhD admissions are entrance-based. Thus, students aspiring to take admission to the programme are required to submit a valid entrance test score. The university accepts the following tests for PhD admissions:
- UGC NET
- CSIR
- ICMR
- GATE
Students submitting a GATE score must note that the they must have cleared the exam within the last two years.
New Question
11 months agoContributor-Level 10
(d) Cs
Size of Cs is the biggest thus, its melting point is the lowest, (d) is correct
New Question
11 months agoContributor-Level 10
1.25 Excess electrons on an oil drop, n = 12
Electric field intensity, E = 2.55
Density of oil, 1.26 g / = 1.26 g/
Acceleration due to gravity, g = 9.81 m/
Charge of an electron, e = 1.60C
Let the radius of the oil drop be r
Force (F) due to electric field (E) is equal to the weight of the oil drop (W)
F = W
Eq = mg
Ene =
= =
r = 9.815 m = 9.815 mm
New Question
11 months agoContributor-Level 10
(i) Lattice energy of BeO is comparatively higher than the hydration energy. Therefore, it is almost insoluble in water. Whereas, BeSO4 is ionic in nature and its hydration energy dominates the lattice energy.
(ii) Both BaO and BaSO4 are ionic compounds but the hydration energy of BaO is higher than the lattice energy therefore it is soluble in water.
(iii) Since the size of Li+ ion is very small in comparison to K+ ion, it polarises the electron cloud of I– ion to a great extent. Thus, Lil dissolves in ethanol more easily than the KI.
New Question
11 months agoContributor-Level 10
After the release of the Woxsen University PhD merit list, selected candidates must present original and self-attested photocopies of documents for verification purposes at the university. Such candidates are required to bring along the following documents:
- Class 10 marksheet
- Class 12 marksheet
- Migration certificate
- School leaving certificate/transfer certificate
- UG marksheets
- UG degree
- PG marksheets (if applicable)
- Letter of experience (if applicable)
- NOC certificate (if applicable)
- Category certificate (if applicable)
- Income certificate (applicable for candidates seeking admission via EWS category)
New Question
11 months agoContributor-Level 10
1.24 The given conditions are explained in the adjacent diagram
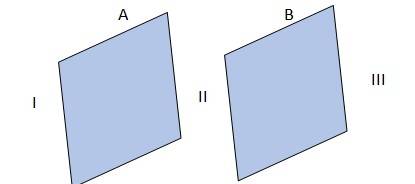
Where A and B represent two large, thin metal plates, parallel and close to each other. The outer surface of A is shown as I, outer surface of B is shown as II and the surface in between A and B is shown as III.
Charge density of plate A, = 17.0 C/
Charge density of plate B, = 17.0
C/
(a) & (b) In the region, I and III, electric field E is zero, because charge is not enclosed by the respective plates.
(c) Electric field, E in the region II is given by
E =
, where
= Permittivity of free space = 8.854
E = N/C = 1.92 N/C
New Question
11 months agoContributor-Level 10
(a) Na2O2 + 2H2O → 2NaOH + H2O2
(b) 2KO2 + 2H2O → 2KOH + O2+ H2O2
(c) Na2O+ CO2 → Na2CO3
New Question
11 months agoContributor-Level 10
In 2024, the TNEA closing rank for BTech in Artificial Intelligence and Data Science at Mohamed Sathak A J College of Engineering rose significantly to 95,591 from 49,987 in 2023, indicating a higher closing rank and increased competition for the programme.
New Question
11 months agoContributor-Level 7
CII Institute of Hospitality, Chandigarh conducts CII Aptitude Test for the selection of right candidates to Diploma programmes. Students must qualify for the test and then appear for an Industry Panel Viva round for the final selection.
New Question
11 months agoContributor-Level 10
(a) Na2CO3 is a salt of a weak acid, carbonic acid (H2CO3) and a strong base NaOH. Thus, it undergoes hydrolysis to produce strong base NaOH and its aqueous solution isalkaline in nature.
Na2CO3 (s) + H2O (l) → 2NaOH
(b) Because the discharge potential of alkali metals is much higher than that of hydrogen, therefore when the aqueous solution of any alkali metal chloride is subjected to electrolysis, H2, instead of the alkali metal, is produced at the cathode. Therefore, alkali metals are prepared by electrolysis of their fused chlorides.
(c) Since potassium is move reactive than sodium and it is found in nature to a less exte
New Question
11 months agoContributor-Level 10
1.23 Electric field produced by the infinite line charge at a distance d having linear charge density
is given by
E = , where
E = Electric field = 9 N/C
= Permittivity of free space = 8.854
d = 2 cm = 0.02 m
Hence, = 9
8.854
0.02 = 10
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts