Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
The success rate for Indian students who pursue MBBS abroad varies significantly depending on the country and institution. For instance, data indicates that the FMGE passing percentage for graduates from countries like Nepal and Georgia is relatively higher, with recent figures showing a 39.6% pass rate for FMGE in December. However, overall statistics reveal that only about 23% of Indian students who graduated from foreign medical colleges cleared the FMGE.
New Question
11 months agoContributor-Level 9
CII Institute of Hospitality ITC Grand, Goa admissions are completely entrance-based. The admission process comprises three levels. Candidates need to go through each step to confirm their seat at CII Institute of Hospitality ITC Grand, Goa. The detailed steps are as follows:
- Application Process: The mode of application is online at CII Institute of Hospitality ITC Grand, Goa. Candidates can apply directly on the college's official website to apply for their desired Diploma programmes.
- Selection Rounds: CII Institute of Hospitality ITC Grand, Goa admissions are divided into two levels. At first, candidates need to qualify for a
New Question
11 months agoGuide-Level 15
Candidates who have passed any degree of the University of Calicut (including degree programmes of SDE/ Open degree programme of SDE, University of Calicut) or that of any other university or institute or courses recognised by Calicut University, UGC or AICTE with at least 50% aggregate are eligible to apply for MBA course at Monti International Institute of Management Studies.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
(b) CO is highly poisonous to living beings because of its ability to block the delivery of oxygen to the organs and tissues.
New Question
11 months agoContributor-Level 10
CII Institute of Hospitality - Taj Hotels, Amritsar offers UG Diploma and Degree in Tourism and Hospitality courses. The selection criteria are based on the CII Aptitude Test and Industry Panel Viva. The candidates must pass Class 12 with a 50% aggregate to enrol for Swiss Professional Diploma course and only Class 12 with for Degree in Tourism and Hospitality course.
New Question
11 months agoContributor-Level 10
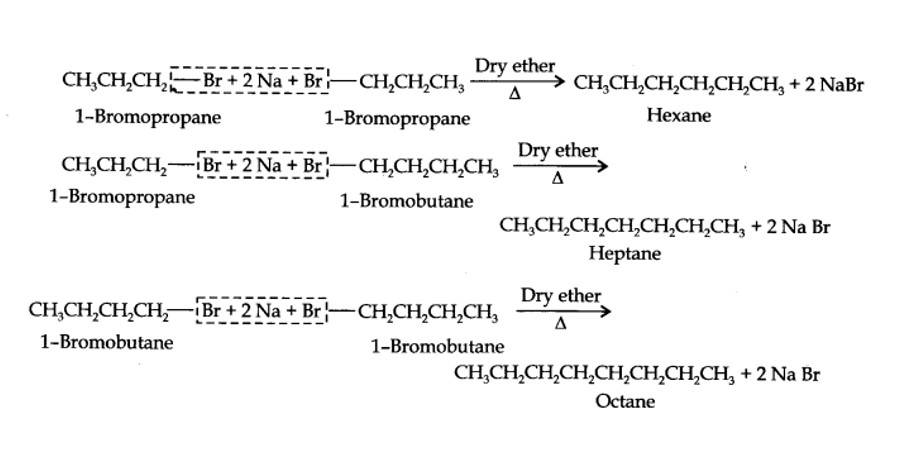
Alkyl halides on treatment with sodium metal in dry ethereal (free from moisture) solution give higher alkanes. This reaction is known as Wurtz reaction and is used for the preparation of higher alkanes containing even number of carbon atoms.
For preparation of alkanes containing odd number of carbon atoms, a mixture of two alkyl halides has to be used. Since two alkyl halides can react in three different ways, therefore, a mixture of three alkanes instead of the desired alkane would be formed. For example, Wurtz reaction between 1-bromopropane and 1-bromobutane gives a mixture of three alkanes i.e., hexane, heptane and octane as shown
New Question
11 months agoContributor-Level 10
3.46. (b) Electronegativity refers to the tendency of an atom to share electrons with another atom.
New Question
11 months agoContributor-Level 10
Anhydrous Ferric Chloride (FeCl3) is another Lewis acid which can be used during ethylation of benzene.
New Question
11 months agoContributor-Level 10
No, candidates looking for course admission at CII Institute of Hospitality - Taj Hotels, Amritsar cannot get direct admission. The institute selects candidates based on CII Aptitude Test and Industry Panel Viva. The eligibility criteria for the course is to pass class 12.
New Question
11 months ago
Contributor-Level 10
US News & World Report considers several factors while determining the rankings of top universities such as graduation rates, graduation performance rates, college graduate salaries, borrower debt, peer assessment, financial resources per student, faculty salaries, standardized tests, etc. The ranking weightage of some of these factors are given as follows:
Stanford University Ranking Criteria (US News) | Weightage |
|---|---|
Average 6-year graduation rate | 16% |
6-year graduation rate of students who received a Pell Grant | 5.5% |
6-year graduation rate of students who did not receive a Pell Grant | 5.5% |
Average first-year student retention rate | 5% |
Over performance (+)/Underperformance (-) | 10% |
New Question
11 months agoContributor-Level 10
Sumandeep Vidyapeeth BSc admission takes place throughmerit secured in Class 12th board exams. Candidates need to have a minimum aggregate of 35% in Class 12 to be eligible for admission in the programme. Here is a step-by-step guide to secure admission in the Sumandeep Vidyapeeth BSc programme:
- Appear for Class 12 board exams
- Apply for admission once results are announced
- Select the specialisation
- Complete Document Verification
- Pay the tuition fee
New Question
11 months agoContributor-Level 9
Yes, CII Institute of Hospitality ITC Grand, Goa conducts an in-house entrance exam for admission to Diploma programmes. To secure a seat to any of the course at the college, students must qualify for CII Aptitude Test, followed by Industry Panel Viva.
New Question
11 months agoContributor-Level 10
CH3 group is electron-donating while -NO2 group is electron-withdrawing. Therefore, maximum electron density will be in toluene, followed by benzene and least in m-dinitrobenzene. Therefore, the ease of nitration decreases in the order: toluene > benzene > m-dinitrobenzene.
New Question
11 months agoContributor-Level 10
Ferromagnetism: The substances that are strongly attracted by a magnetic field are called ferromagnetic substances can be permanently magnetised even in the absence of a magnetic field. Some examples of ferromagnetic substances are iron, cobalt, nickel, gadolinium, and CrO2. In solid state, the metal ions of ferromagnetic substances are grouped together into small regions called domains and each domain acts as a tiny magnet. In an un-magnetised piece of a ferromagnetic substance, the domains are randomly-oriented and so, their magnetic moments get cancelled. However, when the substance is placed in a magnetic field, all the domains get
New Question
11 months agoContributor-Level 10
The MBA sector-wise placement rates at DY Patil Akurdi MBA have been out placement year 2022 and 2023. Check out the table for reference:
Particulars | Placement Statistics (2022) | Placement Statistics (2023) |
|---|---|---|
Marketing | 99% | 92% |
HRM | 86% | 80% |
Finance | 72% | 70% |
Operations | 63% | 78% |
BA | 67% | 50% |
Note- The placements for batch 2024 are ongoing so, the sector-wise reports are yet to be out.
New Question
11 months agoContributor-Level 10
(a) The typical reactions of benzene are electrophilic substitution reactions. Higher the electron-density in the benzene ring, more reactive is the compound towards thesereactions. Since NO2 is a more powerful electron-withdrawing group than Cl, therefore, more the number of nitro groups, less reactive is the compound. Thus, the overall reactivity decreases in the order:Chlorobenzene > p-nitrochlorobenzene > 2, 4-dinitrochlorobenzene
(b) Here, CH3 group is electron donating but NO2 group is electron-withdrawing. Therefore, the maximum electron-density will be in toluene, followed by p-nitrotoluene followed by p-dinitrobenzene. Thus,
New Question
11 months agoContributor-Level 10
The Placement Cell at Dr. D. Y. Patil Institute of Management Studies helps students find jobs. They have a dedicated team that supports students in preparing for employment. This support has led to many students landing good jobs in recent years. The Training & Placement service is available all year to connect students with companies. The Placement Team answers students' questions and provides advice on job applications, resumes, and interview practice for various careers. Their goal is to equip students with the necessary information and skills for a successful job search and to guide those interested in specific career paths.
New Question
11 months agoContributor-Level 10
3.45. (b) It has only ones-electron and hence can be placed in group 1 (alkali metals). It can also gain an electron to achieve a noble gas arrangement and hence it can behave similarly to a group 17 (halogen family) element. Because it is a special case, we shall place hydrogen separately at the top of the Periodic Table
New Question
11 months agoContributor-Level 10
The basic skeleton structure of 2-methylbutane is
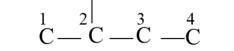
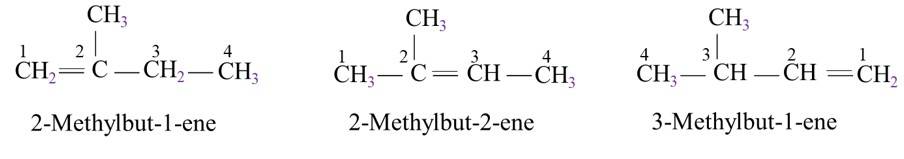
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts