Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
CII Institute of Hospitality - Taj Hotels, Amritsar admissions for all courses are currently ongoing. Interested candidates must apply at the official website. The list of documents mentioned below:
- Scanned Class 10 and Class 12 marksheet
- Leaving Certificate
- Aadhaar Card
- Passport-size photograph
- Signature
New Question
11 months agoContributor-Level 10
There are 510+ private Digital Marketing colleges in India. The table below provides information about the course fees at these top private Digital Marketing colleges in India:
College Name | Average Fees |
|---|---|
INR 90,000 | |
INR 6.4 L - 7.12 Lakh | |
INR 12.54 Lakh | |
INR 9.6 Lakh | |
IMT Ghaziabad - Institute of Management Technology Admission | INR 1.5 Lakh |
Disclaimer: This information is sourced from the official website and may vary.
New Question
11 months agoContributor-Level 10
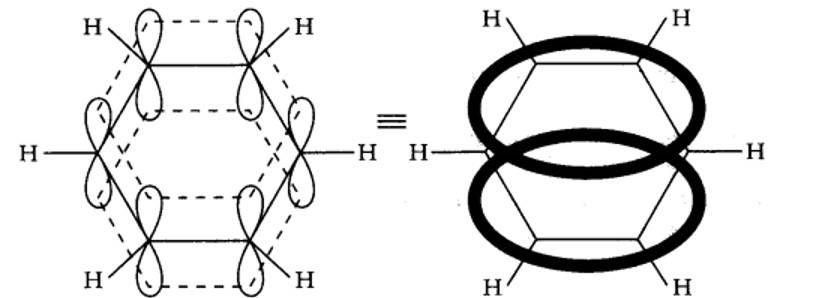
Benzene is a resonance hybrid of two canonical forms. In the resonance hybrid, all the six pi electrons are completely delocalized. This results in resonance stabilization.
New Question
11 months agoContributor-Level 10
CII Institute of Hospitality - Taj Hotels, Amritsar admissions for all courses are currently ongoing. Interested candidates can apply in online mode through the official website. The online application process is explained in the following steps:
Step 1: Visit the official website of CII Institute of Hospitality - Taj Hotels, Amritsar.
Step 2: Fill out the form.
Step 3: Complete the form and submit it.
New Question
11 months agoContributor-Level 10
The structures of cis- and trans-isomer of hex-2-ene are:
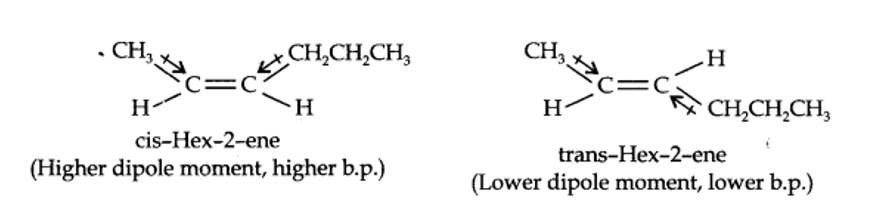
The boiling point of a molecule depends upon dipole-dipole interactions. Since cis-isomer has higher dipole moment, therefore, it has higher boiling point.
New Question
11 months agoContributor-Level 10
1.45 For fcc unit cell, a=2√2?.
Here, a is the edge length and r is the atomic radius (0.144 nm).
a = 2√2 ×0.144 = 0.407 nm
Hence, the length of a side of a cell is 0.407 nm.
New Question
11 months agoGuide-Level 15
Yes, KMAT is one of the accepting exams for admission to MBA programme at Monti International Institute of Management Studies. However, it is not mandatory for aspirants to have KMAT score. Candidates with a valid score in CAT, MAT or CMAT can also get admission in the MBA programme. Once selected, aspirants have to pay the course fee to confirm their seat in the institute.
New Question
11 months agoContributor-Level 10
Digital Marketing colleges in India admit students through various entrance exams. The table below gives some helpful details about the entrance exam dates, schedules, and syllabus.
Exam Name | Exam Date | Exam Schedule | Exam Syllabus |
|---|---|---|---|
March, 2026 (Tentative) | |||
April 2026 (Tentative) | |||
January 2026 (Tentative) | |||
January 2026 (Tentative) |
Disclaimer: This information is sourced from the official website and may vary.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
3.40. Within a period, the oxidising character increases from left to right. Therefore, among F, O and N, oxidising power decreases in the order: F > O > N. However, within a group, oxidising power decreases from top to bottom. Thus, F is a stronger oxidising agent than Cl. Further because O is more electronegative than Cl, therefore, O is a stronger oxidising agent than Cl. Thus, overall decreasing order of oxidising power is: F > O > Cl > N, i.e., option (b) is correct.
New Question
11 months agoContributor-Level 10
A combustion reaction is a reaction in which a substance reacts with oxygen gas, there is a formation of carbon dioxide, water with the evolution of light and heat.
(i) 2C4H10 (g) +13 O2 (g)?8CO2 (g)+10H2O (g) + Heat
(ii) 2C5H10 (g) +15 O2 (g)?10CO2 (g)+10H2O (g) + Heat
(iii) 2C6H10 (g) +17 O2 (g)?12CO2 (g)+10H2O (g) + Heat
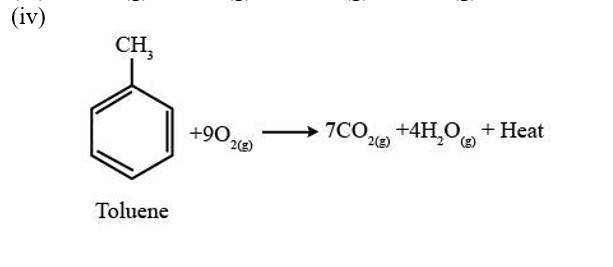
New Question
11 months agoContributor-Level 10
The DY Patil Akurdi MBA has offered decent packages to its MBA graduates. Check the table provided below for more clarity on placement packages between 2022-2024:
Particulars | Placement Statistics (2022) | Placement Statistics (2023) | Placement Statistics (2024) |
|---|---|---|---|
the highest Package | INR 7.80 LPA | INR 10.50 LPA | INR 12 LPA |
Average Package | INR 3.06 LPA | INR 4.18 LPA | INR 4.1 LPA |
New Question
11 months ago
Contributor-Level 10
As per U.S. News ranking, Stanford ranks better than Caltech. Both these universities are the top-ranked universities globally and in the U.S.
University Name | 2024 Ranking | 2025 Ranking |
|---|---|---|
Stanford | 3 | 3 |
CalTech | 23 | 23 |
New Question
11 months agoContributor-Level 10
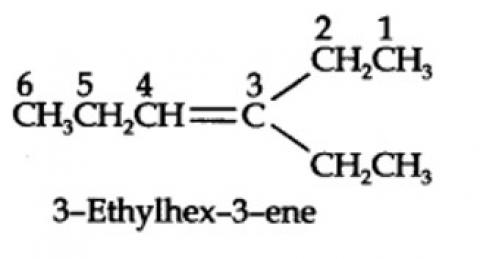
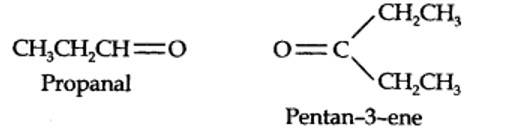
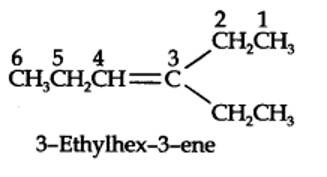
The ozonolysis of 4-Ethylhex-3-ene gives propanal and pentan-3-one.
The structural formula of the alkene (4-Ethylhex-3-ene) is as shown.
New Question
11 months agoContributor-Level 10
1.44 Ge is group 14 element and In is group 13 element. Hence an electron deficient hole is created and therefore, it is p–type.
2. B is group 13 elements and Si is group 14 elements, there will be a free electron. Hence, it is n-type
New Question
11 months agoContributor-Level 10
Yes, Dr. Shakuntala Misra National Rehabilitation University (DSMNRU) does offer scholarships for its BTech programmes. The university offers national as well as state government scholarships through programmes, such as the National Handicapped Finance and Development Corporation and the State Government Scholarship scheme. Students eligible to receive a scholarship can seek these through the university or the corresponding government website.
New Question
11 months agoContributor-Level 10
3.39. In a period, the non-metallic character increases from left to right. Thus, among B, C, N and F, non-metallic character decreases in the order: F > N > C > B. However, within a group, non-metallic character decreases from top to bottom. Thus, C is more non-metallic than Si. Therefore, the correct sequence of decreasing non-metallic character is: F > N > C > B > Si, i.e., option (c) is correct.
New Question
11 months agoContributor-Level 10
The DY Patil Akurdi MBA has had a strong placement record since its inception. Check out the tabulated data given below to know more about DY Patil Akurdi MBA placement in 2022-2024:
Particulars | Placement Statistics (2022) | Placement Statistics (2023) | Placement Statistics (2024) |
|---|---|---|---|
the highest Package | INR 7.80 LPA | INR 10.50 LPA | INR 12 LPA |
Average Package | INR 3.06 LPA | INR 4.18 LPA | INR 4.1 LPA |
Total Recruiters | 52 | 70 | 135 |
Top Recruiters | Reliance, TCS, Bajaj, etc. | TCS, ICICI, Flipkart, etc. | American Express, Bajaj, HDFC Bank, etc. |
New Question
11 months agoContributor-Level 10
(i) An aldehyde with molar mass of 44 u is ethanal, CH3CH=0
(ii) Write two moles of ethanal side by side with their oxygen atoms pointing towards each other.
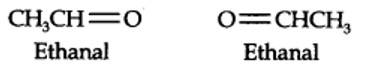
New Question
11 months agoGuide-Level 15
Monti International Institute of Management Studies does not offer direct admission. Candidates are first required to register for admission online. The institute selects candidates for admission to MBA programme based on their score in CAT, MAT, CMAT or KMAT. Selected candidates can confirm their seat by paying the course fee.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
