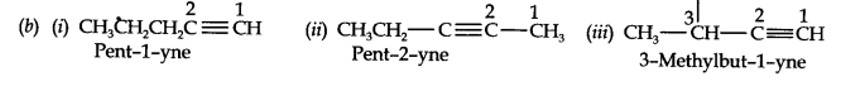Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoNew Question
11 months agoContributor-Level 10
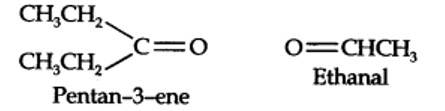
Step 1. Write the structure of the products side by side with their oxygen atoms pointing towards each other.
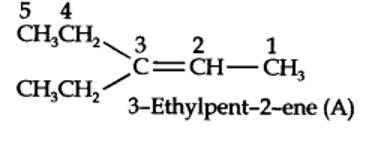
New Question
11 months agoContributor-Level 10
3.38. In a period, metallic character decreases as we move from left to right. Therefore, metallic character of K, Mg and Al decreases in the order: K > Mg > Al. However, within a group, the metallic character, increases from top to bottom. Thus, Al is more metallic than B. Therefore, the correct sequence of decreasing metallic character is: K > Mg > Al > B, i.e., option (d) is correct
New Question
11 months agoContributor-Level 10
1.43 There is one octahedral hole for each atom in hexagonal close packed arrangement. If the number of oxide ions (O2−) per unit cell is 1, then the number of

New Question
11 months agoContributor-Level 10
Sure! Check the table provided below for an overview of the top Digital Marketing colleges in India.
Particulars | Details |
|---|---|
Number of Digital Marketing Colleges in India | 824 colleges |
Annual Fees | Free: 19 colleges Less than INR 1 Lakh: 296 colleges INR 1-2 Lakh: 114 colleges INR 2-3 Lakh: 75 colleges INR 3-5 Lakh: 130 colleges More than INR 5 Lakh: 129 colleges |
Top Digital Marketing Colleges in India | IIM Bangalore, XLRI Xavier School of Management, IIM Calcutta, IIM Indore, IIM Rohtak, IIM Raipur, etc. |
Accepted Entrance Exams | Merit-Based or CAT, MAT, XAT, CMAT, CUET, etc. |
Disclaimer: This information is sourced from the official website and may vary.
New Question
11 months agoContributor-Level 10
RRB Group D answer key is released online within few days after the exam is conducted. Candidates can download their answer keys by logging with the reqquired credentials. Candidates can also raise objections against the answer keys. The answer keys are not made available offline. Candidates need to download it online only.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
A total 135 companies took part in the DY Patil Akurdi MBA placements 2024. Check out the table for reference:
Particulars | Placement Statistics (2022) | Placement Statistics (2023) | Placement Statistics (2024) |
|---|---|---|---|
Total Recruiters | 52 | 70 | 135 |
New Question
11 months agoBeginner-Level 3
- Skillfloor: Skillfloor offers practical Digital Marketing Courses where students learn by doing real projects.They provide live classes, updated tools, and step-by-step guidance from industry experts. Students also get internship opportunities to gain real work experience. Skillfloor supports learners with resume building, interview training, and strong placement help. It is known for its beginner-friendly teaching style and job-focused training approach.
- IIDE (Indian Institute of Digital Education): Provides a strong online and offline curriculum, with a PG-level program, live case studies, 25,000+ real-world campaigns, and very good
New Question
11 months agoContributor-Level 10
There are about 820+ Digital Marketing colleges in India. This includes 510+ private, 40+ government, and 5 public-private colleges. Students can be admitted to the top Digital Marketing colleges in India based on their scores in entrance exams like CAT, MAT, XAT, CMAT, CUET, etc.
New Question
11 months agoContributor-Level 10
1.42 The ratio less than 2:1 in Cu2O shows that some cuprous (Cu+) ions have been replaced by cupric (Cu+2) ions. To maintain electrical neutrality, every two Cu+ ions will be replaced by one Cu+2 ion, thereby creating a hole. As conduction will be due to the presence of these positive holes, hence it is a p -type semi conductor
New Question
11 months agoContributor-Level 10
The highest package offered at DY Patil Akurdi MBA placement in 2024 stood at INR 12 LPA. Refer to the table to know the highest package provided in placement 2022-2024:
Particulars | Placement Statistics (2022) | Placement Statistics (2023) | Placement Statistics (2024) |
|---|---|---|---|
the highest Package | INR 7.80 LPA | INR 10.50 LPA | INR 12 LPA |
New Question
11 months agoContributor-Level 10
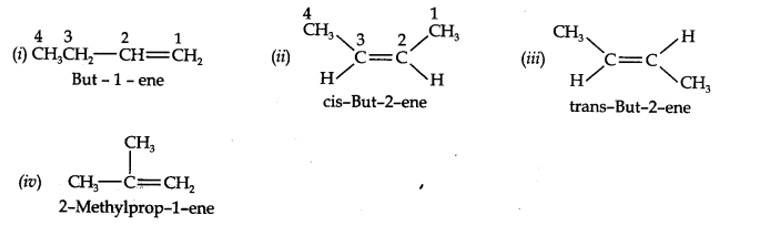
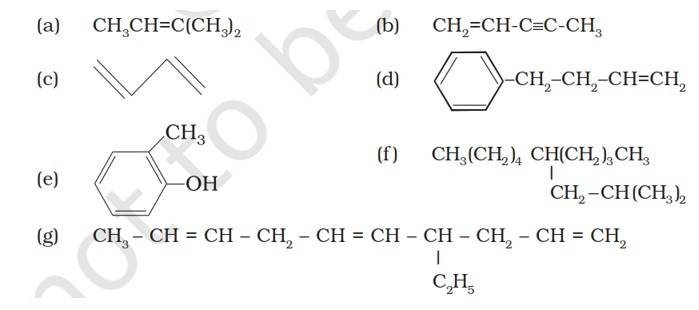
(a) 2-Methykbuut-2-ene
(b) Pent-1-ene-3-yne
(c) But-1,3-diene
(d) 4-Phenylbut-1-ene
(e) 2-Methyl phenol (f) 5- (2-Methylpropyl)decane
(g) 4-Ethyldeca-1,5,8-triene
New Question
11 months agoContributor-Level 10
1.41 These solids have conductive in the intermediate range from 10−6 to 104ohm−1m−1. As there is rise in
the temperature, conductivity also increases because electrons from the valence band jump to
conduction band.
Types of semiconductors
(a) n - type semiconductor when silicon or germanium crystal is doped with group 15 element like P or
As, the dopant atom forms four covalent bonds like a Si or Ge atom but the fifth electron no used in
bonding, becomes delocalised and contribute its share towards electrical conduction. Thus, silicon or
germanium doped with P or As is called n-type semiconduct
New Question
11 months agoContributor-Level 10
Chlorination of methane is a free radical reaction which occurs by the following mechanism involving initiation, propagation and termination steps:

New Question
11 months agoContributor-Level 10
DY Patil Akurdi MBA provides good placements to its graduating students. The students are given excellent placement support every year placing them in top companies. Check out the table below to know the key highlights of the DY Patil Akurdi MBA placement 2022 to 2024:
Particulars | Placement Statistics (2022) | Placement Statistics (2023) | Placement Statistics (2024) |
|---|---|---|---|
the highest Package | INR 7.80 LPA | INR 10.50 LPA | INR 12 LPA |
Average Package | INR 3.06 LPA | INR 4.18 LPA | INR 4.1 LPA |
Total Recruiters | 52 | 70 | 135 |
Top Recruiters | Reliance, TCS, Bajaj, etc. | TCS, ICICI, Flipkart, etc. | American Express, Bajaj, HDFC Bank, etc. |
New Question
11 months agoContributor-Level 10
Admission in Sumandeep Vidyapeeth BSc takes place through the merit secured in Class 12 board exams. To be eligible for admission in the Sumandeep Vidyapeeth BSc candidates need to have a minimum aggregate of 35% in Class 12. If candidates have secured a valid score in Class 12 they can secure admission by applying through the official website. Once selected, candidates will have to pay some amount of the tuition fee to secure their admission into the programme.
New Question
11 months agoContributor-Level 10
Analysis shows that nickel oxide has the formula Ni0.98 O1.00. What fractions of nickel exist as Ni2+ and Ni3+ ions?
1.40 It is given that nickel oxide has the formula as Ni0.98 O1.00.
As per the formula, there are 98 Ni ions for 100 oxide ions.
Out of 98 Ni ions, let x ions be in +2 oxidation state
98−x ions will be in +3 oxidation state.
Oxide ion has −2 charge.
To maintain electrical neutrality, total positive charge on cations = total negative charge on anions.
2x+3(98−x)+100(−2)=0
x=94
Fraction of Ni2+ ions = 94/98 = 0.96
Fraction of Ni2+ ions = 98-94/98 = 0.04
Hence, the fractions of nickel that exists as Ni2+
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts