Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
11.70
Oxidation of propane-1-ol with alkaline KMnO4 solution gives propanoic acid as the product. As the oxidation of primary alcohol gives carboxylic acid as the major product in the presence of a strong oxidizing reagent. And here KMnO4 is a very strong oxidizing agent.
A mixture of o-bromo phenol and p-bromo phenol is formed.
The formation of 2 products depends totally on the reaction conditions.
Dilute HNO3 with phenol.
Only dilute acid will be required for the nitration of phenol, nitric acid contains a small amount of nitrous acid which because of the activation of the ring will be more than enough to nitrate the phenol. Two products
New Question
11 months agoContributor-Level 10
After completing DMLT courses in India, graduates have several options for further career advancement or specialization:
- BSc MLT (Bachelor of Science in Medical Laboratory Technology): A BSc MLT programme typically offers more in-depth study of laboratory techniques, research methods, and advanced topics in medical science.
- Job Opportunities: DMLT course graduates can find employment as medical laboratory technicians or technologists in hospitals, clinics, diagnostic centres, research laboratories, blood banks, and pharmaceutical companies.
- Specialization: After gaining some experience in the field, DMLT course gradua
New Question
11 months agoContributor-Level 10
To secure admission at Navrachana University for BSc (Hons), aspirants must begin with the online registration process. The process is conducted on the university's official website. After the registration/ application process, the university shortlists eligible candidates for further selection rounds. The admission is granted to applicants for various BSc (Hons) specialisations based on all or some of the following selection parameters, depending on the choice of specialisation:
- Class 12 merit
- Statement of Purpose (SOP)
- Interaction with the admissions team
New Question
11 months agoContributor-Level 10
In 2022, Invertis University recorded over 1,500 placements for its BCom students, with more than 300 recruiters participating, resulting in an impressive placement rate of 90%. The highest package offered to a BCom graduate was INR 10 LPA, showcasing the potential of the university's placement support.
New Question
11 months agoContributor-Level 10
The institute offers UG Diploma and Degree in Tourism and Hospitality courses. The selection criteria is based on CII Aptitude Test and Industry Panel Viva. To get admitted to CII Institute of Hospitality - Taj Hotels, Pune, candidates must meet the eligibility criteria set by the institute.
New Question
11 months agoContributor-Level 10
11.69
The –OH group is an electron donating group. Thus, it increases the electron density in the benzene ring as shown by its resonating structure of phenol.
As a result benzene ring is activated towards electrophilic substitution.
New Question
11 months agoContributor-Level 10
11.68
Ortho-nitro phenol is more acidic than ortho-methoxy phenol.
Explanation: Due to strong –R and –I effect of NO2 group, electron density in the O-H bond decreases and hence the loss of a proton becomes easy.
Now after the loss of a proton, the o-nitrophenoxide ion left behind is stabilized by resonance and thus making o-nitro phenol a stronger acid.
In contrast, due to the +R effect of methoxy group increases the electron density in the O-H bond. Thereby making the loss of proton difficult.
Now, the o-methoxyphenoxide ion left after the loss of a proton is destabilized by resonance. The two negative charges repel each other, thereb
New Question
11 months agoContributor-Level 10
GITAM School of Technology Hyderabad appoints expert faculty members and it comprises of PhD, Master's and Diploma holders. The school has well-trained faculty members with years of experience in the related field. The faculty focuses on theoretical and practical knowledge to the students. They engage the students with the latest activities innovative teaching and quality research.
New Question
11 months agoContributor-Level 10
Detroit Mercy University offers 100+ academic courses. Some of the popular majors of this university are Health Professions and Related Programs; Business, Management, Marketing, and Related Support Services; Biological and Biomedical Sciences; Engineering; Architecture-Related Services. Additionally, as per a top-ranking publisher 2025-26 rankings, a few of the graduate programs of this university such as Engineering, Business, Law, Health Professions and Nursing rank among the top in the U.S.
New Question
11 months agoContributor-Level 10
Hi, yes Andhra Pradesh BTech colleges accept JEE Main or AP EAMCET scores. AP EAMCET is the top entrance exam for admission to best colleges for BTech in Andhra Pradesh.
New Question
11 months agoContributor-Level 10
11.67
The acidic nature of phenol can be represented by the following two reactions:-
(a) Phenols react with sodium to give sodium phenoxide, liberating H2.
(b) Phenols react with sodium hydroxide to give sodium phenoxide and water as a by-product.
The acidity of phenol is more than that of ethanol. This is because phenol after losing a proton becomes phenoxide ion which undergoes resonance and is stabilized whereas ethoxide ion does not.
The resonating structures of phenoxide ion are shown as below:
The lone pair of electrons on oxygen delocalizes into the benzene (mesomeric effect) which reduces the electron density in the O-H bond. The
New Question
11 months agoNew Question
11 months agoContributor-Level 10
To get into the UofD MS in Software Engineering, a minimum GPA of 3.0 is required in Bachelor's degree. Also, note that a GPA of 2.7 or higher may be considered with the addition of pre-requisite courses and the approval of the department director / chair and Dean's office. Additionally, the following requirements must also be met:
- Must have taken courses in programming language such as C+ or Java, Data Structures and Algorithms, Calculus, Machine Organization, and Discrete Mathematics
- Also, submit IELTS with a minimum score of 6.5 / TOEFL score of 81 in iBT
New Question
11 months agoContributor-Level 10
9.18. Auto-protolysis means self-ionisation of water. It may be represented as
2H2O(l) + H2O(l) ? H3O+(aq) + OH-(aq)
Acid 1 Base 2 Acid 2 Base 1
Due to auto-protolysis nature of water, it can act as an acid as well as base, i.e. amphoteric in nature.
New Question
11 months agoContributor-Level 10
For Fall 2024, the total number of international students at the University of Detroit Mercy is 5,587. Of which, 3,400+ are UG students, 930+ Graduate students, and 1,200+ Professional students. As per the student diversity, 25% are international students. Out of the total students, 72% are full-time students. In terms of gender enrollment, 57% are women and 43% are men.
New Question
11 months agoContributor-Level 10
The eligibility criteria for Sapthagiri NPS University MBA course is bachelor's degree with a minimum 50% aggregate. Candidates must make sure to have a valid qualification along with the accepted entrance exam scores, KMAT/CAT/MAT/PGCET exams. Further, the university also offers direct admission based on candidates academic performance.
New Question
11 months agoContributor-Level 10
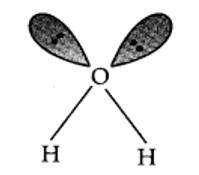
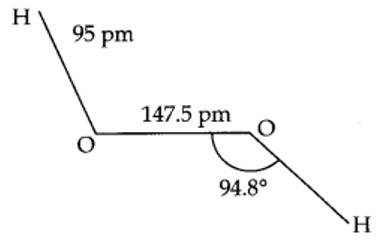
9.17. In water, O is sp3 hybridized. Due to stronger lone pair-lone pair repulsions than bond pair-bond pair repulsions, the HOH bond angle decreases from 109.5° to 104.5°. Thus, water molecule has a bent structure.
New Question
11 months agoContributor-Level 8
SSC Stenographer 2025 vacancies was announced along with the notification. The vacancies are announced category-wise and post-wise. Selection of candidates against the vacancies is done as per the merit-cum-preference. The selection process of SSC Stenographer comprises CBT and skill test.
New Question
11 months agoContributor-Level 9
Here are some important skills that every Communication Design graduate must have for a bright future:
- Technical skills: You must know about Design software and how to use it.
- Creative Thinking: Designers must have a unique and creative approach to attract their targeted audience.
- Communication Skills: They must have good communication skills to make new contacts and build relationships in the industry.
New Question
11 months agoContributor-Level 10
11.66
1. 1-phenylethanol from a suitable - The addition of water takes place according to Markovnikov rule. The alkene taken is styrene. And According to the rule, the positive charge i.e. H+ goes to the carbon of the double bond which has more number of hydrogens and the negative part i.e. OH- goes to the carbon that has less number of hydrogens. Therefore resulting the final product as 1-phenyl ethanol.
2. In the above conversion, NaOH gets dissociated into Na+ and OH-and Na+ then combines with Cl of chloromethylcyclohexane forming NaCl and thus the final product Cyclohexylmethanol is obtained.
3. In this conversion als
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
