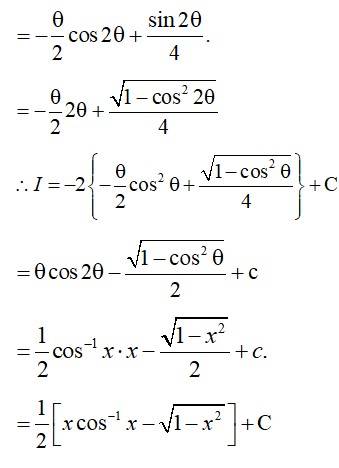Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
The qualities that should be mentioned in an MBA LOR are team skills, leadership abilities, good time management, decision-making capability, the ability to work in challenging situations and diversity, among others.
New Question
10 months agoContributor-Level 10
The institute has not released the average package details as of yet. Check out the table for the highest packages offered to BE CSE between 2023-2025:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
the highest Package | INR 4.7 LPA | INR 4.3 LPA | INR 4.7 LPA |
New Question
10 months agoContributor-Level 10
The composition of any atom can be represented by using the normal element symbol (X) with super-script on the left hand side as the atomic mass number (A) and subscript (Z) on the left hand side as the atomic number (i.e., AZX).
No two elements can have the same atomic number. However, the mass numbers have to be mentioned in order to identify the elements. Thus, symbols 7935Br and 79Br are accepted because atomic number of Br will remain 35 even if not mentioned.
For a given element, the number of protons is the same for the isotopes, whereas the mass number can be different for the given atomic number. Hence, correct pl
New Question
10 months agoContributor-Level 10
You should take an LOR from a teacher/professor/ supervisor/ manager/ someone under whom you have worked. Your recommender should have known you closely for an extended period of time. You cannot take it from your peers or family members.
New Question
10 months agoContributor-Level 10
Heavy metals have a heavy nucleus and contain a large amount of positive change in their nucleus. By using heavy metals like gold and platinum in Rutherford’s experiment, a large number of α-particles get deflected and experience a repulsion thus finding it hard for these α-particles to retrace their path.
If a thin foil of lighter atoms like aluminium were used in the Rutherford’s experiment, the obstruction offered to the path of the fast moving α-particles would be comparatively quite less. As a result, the number of α-particles deflected will be quite less and the particles which are deflected bac
New Question
10 months agoContributor-Level 10
An MS in CS LOR should discuss the various projects and internships taken up by the candidate in the field of computers. Basically, his suitability to pursue an MS in CS should be highlighted by stating if he has knowledge and skills in various domains such as programming, website development, coding, Artificial Intelligence, and others.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
In an LOR for MS Engineering, the suitability of the candidate for the intended programme should be highlighted. This can be done by discussing qualities such as academic prowess, innovation, good analytical skills and critical thinking, the ability to research, an eye for detail, technical skills, and such like.
New Question
10 months agoContributor-Level 10
Charge on oil droplet = – 1.282 x 10-18C
Charge on an electron = – 1.602 x 10-19C
Number of electrons = q /e = (– 1.282 x 10-18C) / (– 1.602 x 10-19C) = 8
New Question
10 months agoContributor-Level 10
The overall BE/BTech placement rate stood at 90% during the Sasurie College of Engineering placements for 2025. Refer to the table for the placement rate recorded between 2023 to 2025:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
Placement Rate | 85% | 90% | 90% |
New Question
10 months agoContributor-Level 10
Static electric charge (q) = 2.5 x 10-16 C
Charge on one electron (e) = 1.602 x 10-19 C
No. of electrons present = (2.5 x 10-16 C) / (1.602 x 10-19 C) = 1560
New Question
10 months agoContributor-Level 9
Yes, it is worth pursuing BTech at Dronacharya Group of Institutions as it is approved by the All-India Council for Technical Education (AICTE) and is aimed at imparting high-quality education in the field of Engineering and Management. It is a leading Engineering College that focuses mainly on innovation, creativity, and excellence in Technical Education. Further, the college offers a BTech programme to students across several specialisations such as Computer Science Engineering, Information Technology, Mechanical Engineering, Electrical and Electronics Engineering, etc.
New Question
10 months agoContributor-Level 10
Let
The integrate is of form,
Comparing the co-efficients,
A + C = 1 .(1)
3A + B + C = 1 .(2)
2A + 2B + C = 1 .(3)
Equation. (2) – 2 × Equation (1),
A + B = –1 .(4)
Equation (3) - (1),
2A + 2B + C = A – C = 1 – 1
A + 2B = 0 .(5)
Equation (5) - Equation (4),
A + 2B - A - B = 0 - (-1)
B = 1.
From (4), A = -1 - B = -1 - 1 = -2.
And from (1), C = 1 - A = 1 - (–2) = 1+2=3
New Question
10 months agoContributor-Level 10
IILET 2026 exam is conducted once a year in an offline mode. The exam is conducted to grant admission to its five-year integrated LLB courses.
New Question
10 months agoContributor-Level 10
(a) The diameter of zinc atom is 2.6 Å =2.6×10−10m.
The radius of Zn atom is (2.6×10−10) / 2=1.3×10−10m=130×10−12m=130 pm.
(b) The number of Zn atoms present on 1.6 cm of length are 1.6 / (2.6×10−8) =6.154×107.
New Question
10 months agoContributor-Level 10
The Sasurie College of Engineering has provided decent BE placements over the years and it is considered worthy. Check out the table below to know the key highlights of the Sasurie College of Engineering placement for BE/BTech during 2023 to 2025:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
the highest Package | INR 4.7 LPA (BE CSE) INR 4.1 LPA (BE ECE) INR 4.7 LPA (BE EE) INR 3.7 LPA (BE ME) INR 4.6 LPA (BE CE) | INR 4.3 LPA (BE CSE) INR 4.4 LPA (BE ECE) INR 4.3 LPA (BE EE) INR 4.3 LPA (BE ME) INR 4.5 LPA (BE CE) INR 4.6 LPA (BTech AIDS) | INR 4.7 LPA (BE CSE) INR 4.5 LPA (BE ECE) INR 4.5 LPA (BE EE) INR 4.5 LPA (BE ME) INR 4.5 LPA (BE CE) INR 4.7 LPA (BTech AIDS) |
the lowest Package | INR 2.7 LPA (BE CSE) INR 2.5 LPA (BE ECE) INR 2.7 LPA (BE EE) INR 2.4 LPA (BE ME) INR 2 LPA (BE CE) | INR 2.3 LPA (BE CSE) INR 2.2 LPA (BE ECE) INR 2.3 LPA (BE EE) INR 2.6 LPA (BE ME) INR 2.4 LPA (BE CE) INR 2.5 LPA (BTech AIDS) | INR 2.7 LPA (BE CSE) INR 2.3 LPA (BE ECE) INR 2.4 LPA (BE EE) INR 2.7 LPA (BE ME) INR 2.7 LPA (BE CE) INR 2.6 LPA (BTech AIDS) |
Placement Rate | 85% | 90% | 90% |
New Question
10 months agoContributor-Level 6
Yes, you must. Never make a second account. It messes up your scores. Log in with your old username. If you forget the password, reset it. The one with duplicate accounts are harder to fix.
New Question
10 months agoContributor-Level 10
The length of the arrangement = 2.4 cm
Total number of carbon atoms present = 2 *108
Diameter of each C-atom = (2.4 cm) / (2 x 108) = 1.2 x 10-8 cm
Radius of each C-atom = ½ x 1.2 x 10-8 cm = 6.0 x 10-9 cm = 0.06 nm
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts